Abstract
Colorectal cancer (CRC) is one of the most common cancers in the World that KRAS mutations are considered as a key step in the progression of CRC. This meta-analysis study aimed to evaluate the prevalence of KRAS mutations in CRC patients in Iran. Six online databases including PubMed, Science direct, Scopus, Web of science, Cochran Library, and Scientific Information Database (SID) were searched systematically up to January 2017. A random-effects meta-analysis was used to calculate the estimation of the prevalence of KRAS mutations in CRC patients by the event rate (ER) with 95% confidence interval (95%CI). Out of 82 articles identified from the search, eleven studies included and analyzed for meta-analysis study. The studies included 1814 CRC patients that mean age of the patients was 57.5 years. The pooled ER of the studies for estimation of the prevalence of KRAS mutation in CRC patients was 32.8% (95%CI=28.7-37.3%). The pooled ER of the studies for the prevalence of codon 12 mutation was 72.5% (95%CI=59.8-82.3%) and for codon 13 mutation was 20% (95%CI=14.6-26.7%). The results showed that the prevalence of KRAS mutation in Iran was different with more studies that therefore the geographical area and race can impact on the prevalence of KRAS mutation in CRC patients. Also, codon 12 had the most prevalence among mutant codons, followed by codon 13 that Gly to Asp and Gly to Val were the most mutations in codon 12.
Introduction
Colorectal cancer (CRC) is the fourth most common cancer in men and the third most common in women Amirifard et al., 2016. There are very few studies about KRAS mutations in CRC from developing countries such as Iran Bishehsari et al., 2006. The latest data from the cancer patients’ registry program showed that the age-standardized incidence rate had risen from 2.8 to 5.5 in 2009 and reached 9.2 in 2012 per 100,000 persons Dolatkhah et al., 2016. KRAS is a proto-oncogene located on the short arm of chromosome 12, encodes the protein KRAS, a GTPase involved in cell division, differentiation and apoptosis Dobre et al., 2015. The prevalence of KRAS mutation in CRC patients is 35–40 %, and the majority of these mutations occur in codon 12 and less frequently in codon 13 of KRAS gene Rosty et al., 2013. Mutations activating the KRAS proto-oncogene are considered a key step in the progression from normal colorectal epithelium to carcinoma Fearon and Vogelstein, 1990. Roughly 90% of the activating mutations, that are influential solitary amino acid replacement in the GTPase pocket that guide to a block of the activity of KRAS p21 protein, are recognized in codons 12 (GGT) and 13 (GGC) of exon 1 and almost 5% in codon 61 (CAA) situated in exon 2. The most regularly found kinds of mutations are G>A and G>T transitions Palmirotta et al., 2009. This meta-analysis study aimed to indicate the prevalence of KRAS mutations in CRC patients in Iran.
Materials and Methods
Search Strategy
Six online databases including PubMed, Science direct, Scopus, Web of science, Cochran Library, and Scientific Information Database (SID) were searched systematically up to January 2017 with the terms of “KRAS” or “K-ras” and “colorectal” or “colon” or “rectum” or “rectal” in combination with “Iran”.
Study selection
One author (E.S) searched the articles and then the second author (M.S) blinded to the first author that if there was any disagreement between two authors, both resolved the problems with two-way conversation. The third author (M.P) did the final revision. The studies were searched for the assessment of prevalence of the KRAS mutations in CRC patients in English abstract.
Data Extraction
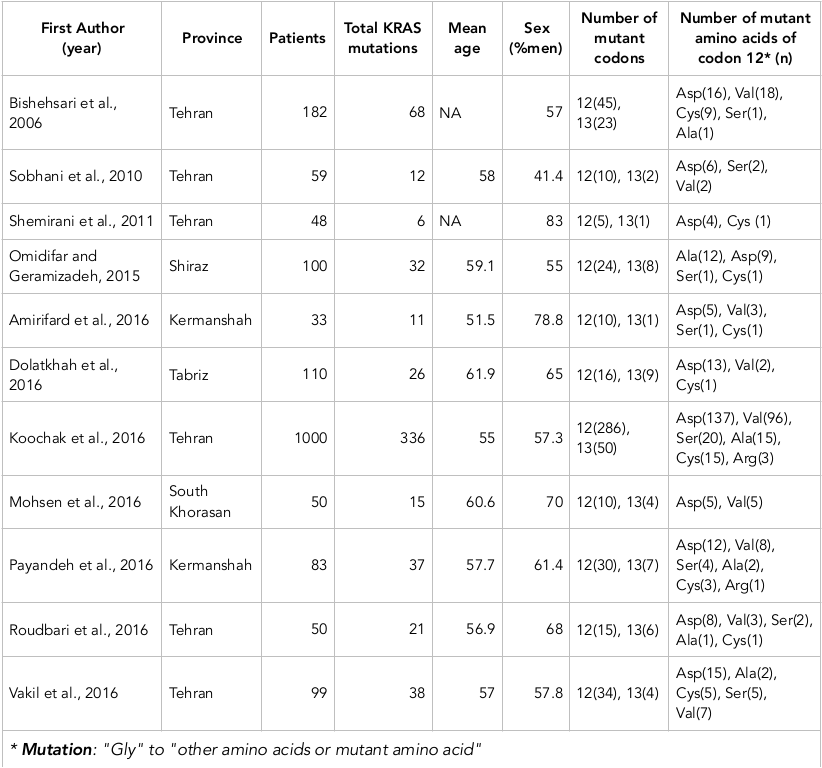
Name of the first author, year of publication, Province of the region, number of CRC patients, number of mutations, percentage of male (%), the mean age, number of mutant codons, and number of mutant amino acids of codon 12 were the relevant data extracted from every study.
Statistical analysis
A random-effects meta-analysis was used by Comprehensive Meta-Analysis software version 2.0 (CMA 2.0). The event rate (ER) with 95% confidence interval (95%CI) was calculated for estimation of the prevalence of KRAS mutations in CRC patients. Heterogeneity between estimates was assessed by the Q and I2 statistic that for the Q statistic, heterogeneity was considered for P<0.1. P-value (2-sided) <0.05 was considered to be statistically significant in this meta-analysis study. Also, publication bias was assessed through funnel plot analysis with the Begg’s and Egger’s tests.
Results
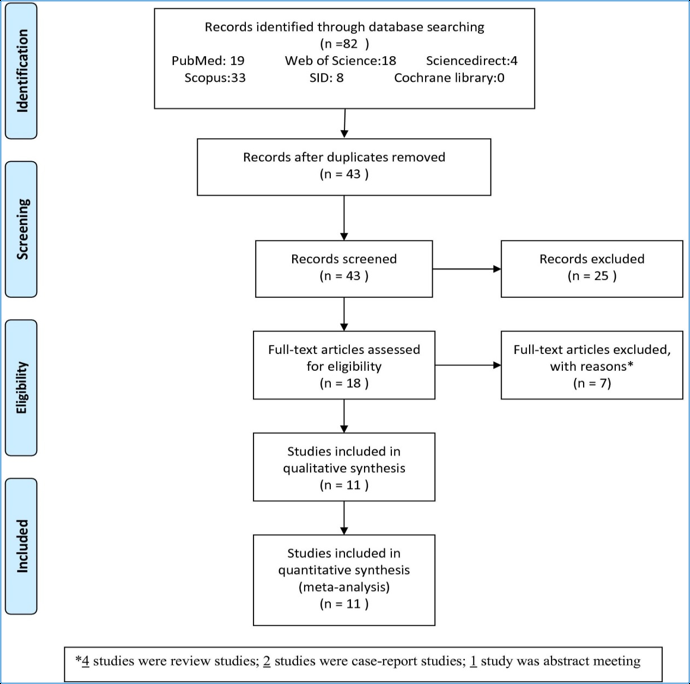
From the initial 82 articles identified from the search, after excluding the studies, 18 studies were assessed for eligibility. Then, seven studies were excluded based on reasons. At last, eleven studies included and analyzed for meta-analysis study ( Figure 1 ).
The characteristics of the studies included in the meta-analysis are shown in Table 1 . All studies are case-control studies published between 2006 and 2016. Six studies were studied from Tehran (Central of Iran) Bishehsari et al., 2006Roudbari et al., 2016Shemirani et al., 2011Sobhani et al., 2010Tameshkel et al., 2016Vakil et al., 2016, one study from Shiraz (Southwest of Iran) Omidifar et al., 2015, two studies from Kermanshah (Western of Iran) Amirifard et al., 2016Payandeh et al., 2016, one study from Tabriz (Northwestern of Iran) Dolatkhah et al., 2016, and one study from South Khorasan (Eastern of Iran) Naseri et al., 2016. All studies included 1814 CRC patients that mean age of the patients was 57.5 years. In more studies, the prevalence of CRC in males was more than females Amirifard et al., 2016Bishehsari et al., 2006Dolatkhah et al., 2016Naseri et al., 2016Omidifar et al., 2015Payandeh et al., 2016Roudbari et al., 2016Shemirani et al., 2011Tameshkel et al., 2016Vakil et al., 2016. Number of mutations in codon 12 and 13 and also mutant amino acid in codon 12 has been shown in Table 1 that Gly is normal amino acid in codon 12 and can change to other amino acids.
KRAS mutation
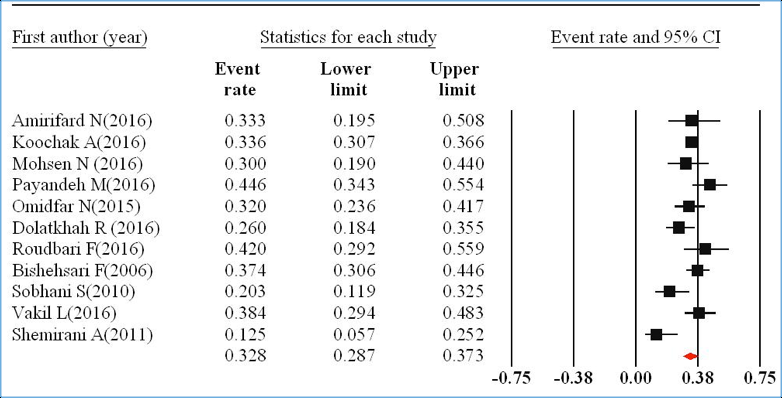
The prevalence of KRAS mutation in CRC patients has been reported in Figure 2 by the ER. The pooled ER of the studies was 32.8% (95%CI=28.7-37.3%) with I2=58.26% (P=0.008).
Mutant codons
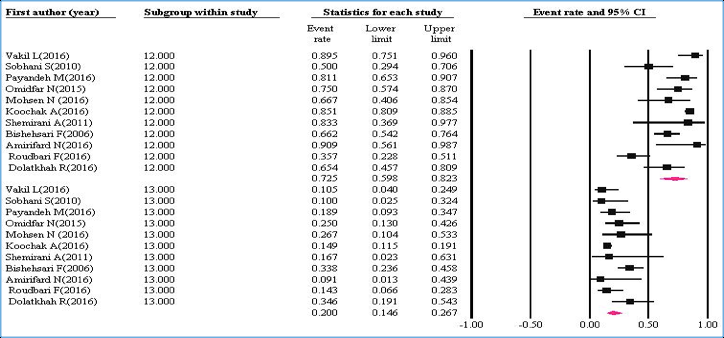
Figure 3 shows the prevalence of mutant codons among all KRAS mutations.
The pooled ER of the studies for codon 12 was 72.5% (95%CI=59.8-82.3%) with I2=83.3% (P<0.001) and for codon 13 was 20% (95%CI=14.6-26.7%) with I2=54.74% (P=0.015).
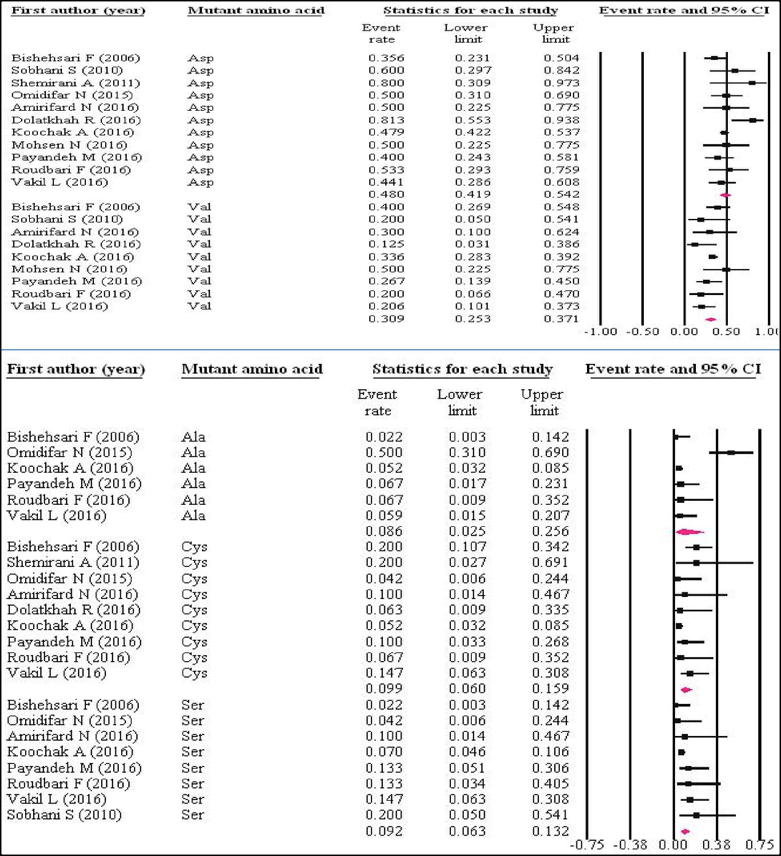
Mutant amino acids of codon 12
Figure 4 shows the prevalence of mutant amino acids among KRAS mutations in codon 12. The pooled ER of the studies for "Asp" was 48% (95%CI=41.9-54.2%), "Val" was 30.9% (95%CI=25.3-37.1%), "Ala" was 8.6% (95%CI=2.5-25.6%), "Cys" was 9.9% (95%CI=6-15.9%), and "Ser" was 9.2% (95%CI=6.3-13.2%).
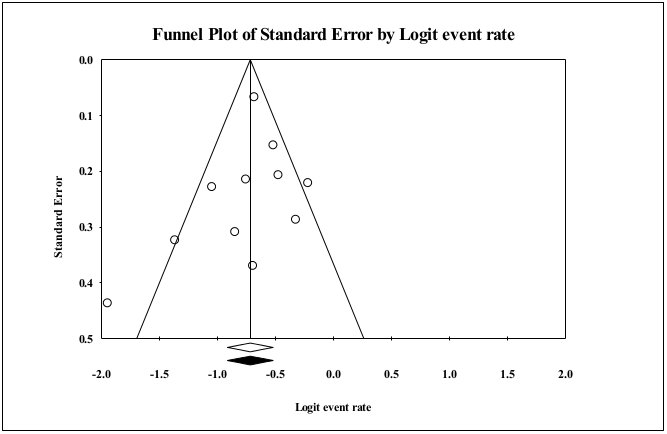
Publication bias
Funnel plot of random effect of the studies for the prevalence of KRAS mutation in CRC patients has been shown in Figure 5 . The Begg’s and Egger’s tests didn’t show publication bias (P=0.102 and P=0.433, respectively).
Limitations
More studies were not sex-matched.
Some studies reported the prevalence just in metastatic CRC and rest of studies both metastatic and non-metastatic CRC.
The studies reported in different areas and races of Iran that geographical area and race can effect on the prevalence of mutations.
In a number of studies, except for codon 12 and 13, types of other different codons have been checked.
Conclusion
The meta-analysis showed that the prevalence of KRAS mutation in Iran was different with more studies that therefore the geographical area and race can impact on the prevalence of KRAS mutation in CRC patients. Also, Codon 12 had the most prevalence among mutant codons, followed by codon 13. At last, Gly to Asp and Gly to Val were the most mutations in codon 12.
Abbreviations
CI: Confidence Interval
CRC: Colorectal Cancer
ER: Event Rate
SID: Scientific Information Database
Author Contributions
Conceptualization: MP ES MS
Data curation: ES MS
Formal analysis: ES MS
Funding acquisition: ES MS
Investigation: NA BS NF
Methodology: ES MS
Project administration: MP NA BS NF
Resources: MP MD
Software: ES MS
Supervision: MP NA
Validation: MP ES MS
Visualization: NA BS NF
Writing – original draft: ES MS
Writing – review & editing: MP NA MS BS NF ES MD
References
-
N.
Amirifard,
E.
Sadeghi,
N.
Farshchian,
A.
Haghparast,
M.
Choubsaz.
Evaluation of KRAS Gene Mutations in Metastatic Colorectal Cancer Patients in Kermanshah Province. Asian Pacific journal of cancer prevention: APJCP.
2016;
17
:
3085-3088
.
-
F.
Bishehsari,
M.
Mahdavinia,
R.
Malekzadeh,
F.
Verginelli,
T.
Catalano,
M.
Sotoudeh,
V.
Bazan,
V.
Agnese,
D.
Esposito,
L.
De Lellis.
Patterns of K-ras mutation in colorectal carcinomas from Iran and Italy (a Gruppo Oncologico dell'Italia Meridionale study): influence of microsatellite instability status and country of origin. Annals of oncology.
2006;
17
:
vii91-vii96
.
-
R.W.
Burt.
Colon cancer screening. Gastroenterology.
2000;
119
:
837-853
.
-
A.S.
Chretien,
A.
Harlé,
M.
Meyer-Lefebvre,
M.
Rouyer,
M.
Husson,
C.
Ramacci,
V.
Harter,
P.
Genin,
A.
Leroux,
J.L.
Merlin.
Optimization of routine KRAS mutation PCR-based testing procedure for rational individualized first-line-targeted therapy selection in metastatic colorectal cancer. Cancer medicine.
2013;
2
:
11-20
.
-
F.
Ciardiello,
S.
Tejpar,
N.
Normanno,
D.
Mercadante,
T.
Teague,
B.
Wohlschlegel,
E.
Van Cutsem.
Uptake of KRAS mutation testing in patients with metastatic colorectal cancer in Europe, Latin America and Asia. Targeted oncology.
2011;
6
:
133
.
-
M.
Dobre,
D.E.
Dinu,
E.
Panaitescu,
R.D.
Birla,
C.-I.
Iosif,
M.
Boeriu,
S.
Constantinoiu,
R.N.
Ivan,
C.M.
Ardeleanu,
M.
Costache.
Kras gene mutations-prognostic factor in colorectal cancer. Rom J Morphol Embryol.
2015;
56
:
671-678
.
-
R.
Dolatkhah,
M.H.
Somi,
I.A.
Kermani,
M.
Bonyadi,
B.
Sepehri,
K.
Boostani,
S.
Azadbakht,
N.
Fotouhi,
F.
Farassati,
S.
Dastgiri.
association between proto-oncogene mutations and clinicopathologic characteristics and overall survival in colorectal cancer in east azerbaijan, iran. OncoTargets and therapy.
2016;
9
:
7385
.
-
Y.A.
El Kader,
G.
Emera,
E.
Safwat,
H.A.
Kassem,
N.M.
Kassem.
The KRAS StripAssay for detection of KRAS mutation in Egyptian patients with colorectal cancer (CRC): A pilot study. Journal of the Egyptian National Cancer Institute.
2013;
25
:
37-41
.
-
E.R.
Fearon,
B.
Vogelstein.
A genetic model for colorectal tumorigenesis. Cell.
1990;
61
:
759-767
.
-
U.
Miglio,
R.
Mezzapelle,
A.
Paganotti,
S.
Allegrini,
C.
Veggiani,
J.
Antona,
S.
Gentilli,
G.
Monga,
O.
Alabiso,
R.
Boldorini.
Mutation analysis of KRAS in primary colorectal cancer and matched metastases by means of highly sensitivity molecular assay. Pathology-Research and Practice.
2013;
209
:
233-236
.
-
M.
Naseri,
A.
Sebzari,
F.
Haghighi,
F.
Hajipoor,
F.E.
Razavi.
Frequency of K-RAS and N-RAS Gene Mutations in Colorectal Cancers in Southeastern Iran. Asian Pacific Journal of Cancer Prevention.
2016;
17
:
4511-4515
.
-
N.
Omidifar,
B.
Geramizadeh,
M.
Mirzai.
K-ras mutation in colorectal cancer, a report from southern Iran. Iranian journal of medical sciences.
2015;
40
:
454
.
-
F.
Ozen,
S.
Ozdemir,
E.
Zemheri,
G.
Hacimuto,
F.
Silan,
O.
Ozdemir.
The proto-oncogene KRAS and BRAF profiles and some clinical characteristics in colorectal cancer in the Turkish population. Genetic testing and molecular biomarkers.
2013;
17
:
135-139
.
-
R.
Palmirotta,
A.
Savonarola,
V.
Formica,
G.
Ludovici,
G.
Del Monte,
M.
Roselli,
F.
Guadagni.
A novel K-ras mutation in colorectal cancer. A case report and literature review. Anticancer research.
2009;
29
:
3369-3374
.
-
M.
Payandeh,
B.
Shazad,
M.
Sadeghi,
M.
Shahbazi.
Correlation between RAS test results and prognosis of metastatic colorectal cancer patients: a report from Western Iran. Asian Pac J Cancer Prev.
2016;
17
:
1729-1732
.
-
C.
Rosty,
J.P.
Young,
M.D.
Walsh,
M.
Clendenning,
R.J.
Walters,
S.
Pearson,
E.
Pavluk,
B.
Nagler,
D.
Pakenas,
J.R.
Jass.
Colorectal carcinomas with KRAS mutation are associated with distinctive morphological and molecular features. Modern Pathology.
2013;
26
:
825
.
-
F.
Roudbari,
B.
Poopak,
F.
Sheikhsofla,
M.
Ghadiani.
Evaluation of frequency of kirsten rat sarcoma gene mutations in Iranian colorectal cancer. Tehran University Medical Journal TUMS Publications.
2016;
74
:
392-399
.
-
F.
Selcukbiricik,
S.
Erdamar,
C.
Ozkurt,
N.
Molinas Mandel,
F.
Demirelli,
M.
Ozguroglu,
D.
Tural,
E.
Buyukunal,
S.
Serdengecti.
The role of K-RAS and B-RAF mutations as biomarkers in metastatic colorectal cancer. J BUON.
2013;
18
:
116-123
.
-
A.I.
Shemirani,
M.M.
Haghighi,
S.
Milanizadeh,
M.Y.
Taleghani,
S.R.
Fatemi,
B.
Damavand,
Z.
Akbari,
M.R.
Zali.
The role of kras mutations and MSI status in diagnosis of colorectal cancer. Gastroenterology and Hepatology from bed to bench.
2011;
4
:
70
.
-
S.
Sobhani,
M.
GHAFFARPOUR,
H.Z.
MOSTAKHDEMIN,
F.
Kamali,
M.Z.
NOUR,
M.
HOUSHMAND.
The prevalence of common mutation frequency in K-ras codons 12, 13 in Iranian Colorectal Cancer patients. 2010
.
-
E.
Symvoulakis,
A.
Zaravinos,
D.
Panutsopulos,
O.
Zoras,
E.
Papalambros,
F.
Sigala,
D.
Spandidos.
Highly conserved sequence of exon 15 BRAF gene and KRAS codon 12 mutation among Greek patients with colorectal cancer. International Journal of Biological Markers.
2007;
22
:
12
.
-
F.S.
Tameshkel,
M.R.
Sohrabi,
M.
Reza,
H.R.
Babaee,
B.
Bahar,
F.
Imanzade,
F.
Zamani,
M.R.
Khonsari,
H.
Ajdarkosh,
G.
Hemmasi.
Mutation analysis of KRAS and BRAF genes in metastatic colorectal cancer: a first large scale study from Iran. Asian Pacific Journal of Cancer Prevention.
2016;
17
:
603-608
.
-
L.
Vakil,
R.
Najafipour,
N.
Rakhshani,
F.
Zamani,
A.
Morakabati,
A.
Javadi.
Investigation of FIH-1 and SOCS3 expression in KRAS mutant and wild-type patients with colorectal cancer. Tumor Biology.
2016;
37
:
8841-8848
.
Comments
Downloads
Article Details
Volume & Issue : Vol 4 No 10 (2017)
Page No.: 1693-1704
Published on: 2017-10-16
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 9051 times
- Download PDF downloaded - 2459 times
- View Article downloaded - 5 times
 Biomedpress
Biomedpress