Therapeutic use of fisetin, curcumin, and mesoporous carbaon nanoparticle loaded fisetin in bleomycin-induced idiopathic pulmonary fibrosis
Abstract
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive and lethal lung disorder of unknown etiology Cottin et al., 2014Kondoh et al., 1993Raghu et al., 2006. IPF primarily occurs between 60 and 70 years of age, and is slightly more predominant in males Gribbin et al., 2006Raghu et al., 2006. Data from around the world demonstrates that IPF favors no particular race or ethnic group. IPF is considered a complex disease where both genetic and environmental factors are believed to contribute to disease susceptibility Kondoh et al., 1993. The disease appears to be driven by abnormal and/or dysfunctional alveolar epithelial cells (AECs) that promote fibroblast recruitment, proliferation, and differentiation, resulting in scarring of the lung, architectural distortion, and irreversible loss of function King et al., 2011. Exerciseinduced breathlessness and chronic dry cough are the prominent symptoms. The onset of symptoms is slow, but symptoms become progressively worse over time. The initial presentation of breathlessness is commonly attributed to aging, cardiac disease, or emphysema which results in typical delays of diagnosis. At the cellular level, IPF is characterized by alveolar epithelial injury, initiation of inflammatory cascades, exaggerated pro-fibrotic cytokine expression, increased extracellular matrix (ECM) deposition, and the development of fibrotic lesions known as fibroblast “foci” Selman et al., 2001. Injured epithelium could release growth factors, cytokines and matrix metalloproteinase, which caused the activation or proliferation of mesenchymal cell, deposition of extracellular matrix and the accumulation of fibroblasts (Yan et al., 2014). The disease progresses towards chronic restrictive respiratory failure and death Cottin and Cordier, 2012Raghu et al., 2011a. IPF is associated with a median survival of only 3–5 years following diagnosis Collard et al., 2003. In their study, Japanese physicians were the first to describe acute, unexpected deterioration in patients with IPF. This phenomenon has been called the "acute exacerbation" or, more euphemistically, the "terminal complication" of IPF Kondoh et al., 1993.
IPF belongs to a family of lung disorders known as the interstitial lung diseases (ILD) or, more accurately, the diffuse parenchymal lung diseases (DPLD). Within this broad category of diffuse lung diseases, IPF belongs to the subgroup known as idiopathic interstitial pneumonia (IIP). During IPF, airway remodelling occurs which can be defined as changes in the composition, content, and organization of the cellular and molecular constituents of the airway wall (Barratt and Millar, 2014;Kondoh et al., 1993).
The pathogenesis of the disease remains poorly understood, although current paradigms focus on the importance of alveolar epithelial cell injury as a critical initiating event with subsequent dysregulated wound healing and fibrosis, resulting in distortion of the lung architecture. The activation of cell-signaling pathways through tyrosine kinases such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), and platelet-derived growth factor (PDGF) has been implicated in the pathogenesis of the disease Chaudhary et al., 2007Coward et al., 2010Wollin et al., 2014. IPF not only destroys the normal lung parenchyma but also affects the pulmonary vasculature with aberrant microvascular and macrovascular remodeling (Barratt and Millar, 2014). Previous studies showed that IPF develops from chronic epithelial cell injury and aberrant activation of progressive fibrosis Selman et al., 2001. Therefore, the therapeutic strategy against IPF has shifted from corticosteroids and/or immunosuppressants to antifibrotic agents King et al., 2008Taniguchi et al., 2010.
Current medical therapy for IPF is poorly effective. However, IPF is a progressive, ultimately fatal disorder for which substantive medical therapy is desperately needed. The only care options endorsed by guidelines published in 2011 were pulmonary rehabilitation, long-term oxygen therapy, lung transplantation, and enrolment in a clinical trial Raghu et al., 2011b.
Animal models play an important role in the investigation of diseases, and many models are established to examine pulmonary pathobiology. Chronic diseases such as IPF are more difficult to model, since the etiology and natural history of the disease is unclear and no single trigger is known that is able to induce IPF in animals. Different models of pulmonary fibrosis have been developed over the years. Common methods include radiation damage, instillation of bleomycin, silica or asbestos and transgenic mice or gene transfer employing fibrogenic cytokines. So far, the standard agent for induction of experimental pulmonary fibrosis in animals is bleomycin Moeller et al.,2008.
Bleomycin (bleo) is a glycopeptide-derived antibiotic, isolated from the soil fungusStreptomyces verticillus. It is a chemotherapeutic agent with side effects especially on skin. The most serious complication of bleomycin is pulmonary fibrosis and impaired lung function. It has been suggested that bleomycin induces sensitivity to oxygen toxicity and some studies support the role of the proinflammatory cytokines IL-18 and IL-1β in the mechanism of bleomycin-induced lung injury Hoshino et al., 2009. Animal models are often used to investigate pulmonary fibrosis, and they play an important role in understanding the pathogenesis of this disease. Bleomycin model is the most widely used animal model of pulmonary fibrosis. It is widely used as an inducer in the animal models Manali et al., 2011Moeller et al., 2008. Lung fibrosis induced by bleomycin delivered to animals via different routes has different pattern of foci distributions. Using the bleomycin-induced pulmonary fibrosis model, it has been previously reported that pulmonary inflammation and fibrosis are mediated by secretion of the proinflammatory and pro-fibrotic cytokine IL-1β through Nlrp3 inflammasome activation and IL-1R1/MyD88 signaling Chen et al., 2015Francois et al., 2015Gasse et al., 2007. Fibrosis associated with bleomycin treatment has also been linked to toxic reactive oxygen and nitrogen species produced by infiltrating inflammatory cells Yamazaki et al., 1998. Thus, agents that depress oxidative stress are also of potential clinical value, and could have additional protective effects against bleomycin-induced pulmonary fibrosis.
Plant polyphenols are a class of molecules characterized by the presence of multiple phenol groups in their structural moiety. Over the past several years, polyphenols have been studied for their potential to modulate the production and activity of inflammatory molecules Quideau et al., 2011. Fisetin (3, 7, 3′, 4′- tetrahydroxyflavone) is a flavonol, a structurally distinct chemical substance that belongs to the flavonoid group of polyphenols found in many plants fruits and vegetables, such as strawberries, apples, persimmons, onions and cucumbers. Fisetin inhibits the activity of several pro-inflammatory cytokines, including tumor necrosis factor alpha, interleukin 6, and Nuclear factor kappa B Gupta et al., 2014Sahu et al., 2014. In addition, fisetin is a potent natural anticancer agent.
The polyphenol curcumin is the active ingredient in the herbal remedy and dietary spice turmeric. Chemically, curcumin exhibits keto–enol tautomerism having a predominant keto form in acidic and neutral solutions and stable enol form in alkaline medium (Anand et al., 2007). This vibrant yellow spice, derived from the rhizome of the plantCurcuma longa, has a long history of use in traditional medicines of China and India, where it is used to treat inflammatory diseases, abdominal disorders, and a variety of other ailments (Ammon and Wahl, 1991). Curcumin has been shown to exhibit antioxidant, anti-inflammatory, antiviral, antibacterial, antifungal, and anticancer activities and thus has a potential against various malignant diseases, diabetes, allergies, arthritis, Alzheimer’s disease, and other chronic illnesses Aggarwal,2007maManolova et al., 2014. Extensive scientific research over the past decade has shown the ability of this compound to modulate multiple cellular targets and hence possesses preventive and therapeutic value against a wide variety of diseases (Anand et al., 2007). Curcumin has been shown to suppress TNF expression in in vitro, in vivo,and human studies. It can suppress TNF expression induced by numerous stimuli and in numerous cell types. Recent work has suggested that curcumin acts as a cancer chemopreventive and chemotherapeutic agent Chuang et al., 2000Goel et al., 2008Hatcher et al., 2008Jiao et al., 2009. Curcumin inhibits activation of nuclear factor κB through blockade of IκB kinase, and inhibits activation of cyclooxygenase 2 (COX2). It also alters activator protein 1 (AP1) complexes and inhibits Akt. In addition to the effects on transcription and cell signaling, curcumin possesses chemical features that may further modulate its chemopreventive activity Deeb et al., 2007Jobin et al., 1999Woo et al., 2003.
In the light of the promising properties and broad spectrum of activities of fistine and curcumin, the aim of our study was to evaluate the beneficial effect of fistine, curcumin and mesoporous carbaon nanoparticle (MCN) loaded fisetin as an anti-inflammatory agents against bleomycin-induced changes in mice with idiopathic pulmonary fibrosis.
Material and methods
Mice
Balb/C mice from national institute of nutrition (NIN) Hyderabad were used. The following numbers of animals used in each group were as follows: a) control [3], b) bleomycin treated [4], C) bleomycin + fisetin treated [4], D) bleomycin + curcumin treated [4], E) bleomycin +fisetin + mesoporous carbon nanoparticle treated [4] . Total 19 animals were maintained under pathogen free condition and given food and water routinely. All experiments were performed according to rules laid down by the Institutional and departmental animal ethics committee and the animals housed under specific pathogen-free conditions at the animal house of the department of Zoology, University of Calcutta.
Treatment of mice
Treatment was performed with adult Balb/C mice. 16 mice were anaesthetized using Propofol received a single dose of bleomycin (naprobleo, miracalus) both intratracheally and intranasally, bleomycin 0.075U/ml bleomycin dissolved in 1ml of 0.09% sterile saline water, from here 16 mice received a single dose of bleomycin at day 0.20μl was administered intranasally and 40μl from intratracheally. Mice were administered with 40μl of fisetin, 40μl of curcumin and 40μl mesoporous carbon nano (MCN) particle conjugated fisetin intratracheally at day 7, 14 and 21, and at the 28 th day they were sacrificed, organ such as lung, bone marrow, liver, bronchoalveolar lavage, and peripheral blood were collected. Weight and other parameter of mice were taken on a regular basis according to rodent health monitering programme (RHMP).
Mice developed marked interstitial and alveolar fibrosis, collagen content was detected by Hydroxyproline estimation, blood smear preparation and cytospin sample staining with hematoxylin was used to quantify the differential cell count of blood, clonogenic potential was detected by colony forming unit (CFU) assay, cytokine analysis of Balf and peripheral blood was used to study the cytokine profile, weight of mice was taken in a regular basis to observe the development of fibrosis.
Colony Forming Unit (CFU)-c assay
For quantification of committed progenitors of all lineages, Colony Forming Units in culture (CFU-c) were performed using standard protocol. Briefly, after dissection, the tissues (Spleen, Lung) were immediately kept in Iscove’s Modified Dulbecco’s Media (IMDM) (purchased from Himedia, India) maintaining aseptic conditions. For Bone marrow samples, the bone was taken into the Biosafety cabinet (Vision Scientific, Korea) and flushed with PBS until the bone turns white. The cell suspension was then kept in IMDM. The tissues were minced and the cell suspension was collected with the help of a nylon mesh. Spleen and lung samples were centrifuged at 5000 rpm for 5 minutes. Bone marrow was centrifuged at 5000 rpm for 10 minutes. Cell count was taken. Number of cells per well taken was 1x104 . For bone marrow, 4x104 cells were taken per well. CFU-c media was prepared using IMDM, supplemented with 30% FBS (Himedia, India), 10% BSA (Biosera), 1% Penicillin-Streptomycin (Himedia, India) and 5 ng/ml murine SCF (Biovision). Lastly, 1.5% methylcellulose (in powdered form purchased from Himedia, India) was added into the concoction. 1ml CFU-c assay media and 500μl cell suspension was plated in each 24 well cell culture plate. The 24 well plate (NEST Biotech Co.Ltd.) was kept in CO2 incubator (Thermo Scientific) at 5%CO2 and 37°C for 14 days. All Colony types were counted using Floid Cell Imaging Station (Life Technologies, India.) and pooled to get total CFU-c.
Total Count/Differential Count
White blood cells (WBCs) are a heterogeneous group of nucleated cells. They play an important role in phagocytosis and immunity. Differential white blood cell count is an examination and enumeration of the distribution of leukocytes in a stained blood smear. The different kinds of white cells are counted and reported as percentages of the total WBCs examined. Increases in any of the normal leukocyte types or the presence of immature leukocytes or erythrocytes in peripheral blood are important diagnostically in a wide variety of inflammatory disorders. Differential count was taken using the following protocol. Blood was collected in 1X RBC lysis buffer. It was kept at room temperature for 5 minutes, then flushed with 1X PBS [137 mM NaCl (Merck), 2.7 mM KCl (Himedia), 10 mM Na2HPO4 (Qualigens), 2 mM KH2PO4 (Himedia)], and centrifuged (Cold Centrifuge Vision VS- 15000CFN) at 4000 rpm for 5 minutes at 4°C. The supernatant was discarded. The pellet is dissolved in 1X PBS and stored at 4°C. 105 cells, dissolved in PBS, were diluted in cold 1% BSA-PBS. Quickly, 100 μl of each sample was added to appropriate wells of the cytospin (Centurion Scientific), and the slides and filters were placed in the correct slots of the cytospin. The slides were centrifuged at 2000 rpm for 3 minutes. The slides were removed and air dried. They were then fixed with methanol (SRL), and air dried before staining. The fixed slides were placed in 100% Ethanol in a Coplin Jar for 5 minutes, followed by 10 minutes in 90% ethanol. They were then stained with hematoxylin for 5 minutes, rinsed in 70% ethanol, counterstained with eosin for 2 minutes, and again rinsed in 70% ethanol. Then they were placed in 100% ethanol for 1 minute, and then observed under the microscope (Debro DX-200).
Collagen content estimation by hydroxyproline assay
Collagen is the major structural protein of the cellular matrix of the lung. The most common method for evaluating tissue fibrosis/collagen deposition is hydroxyproline quantification. 4-Hydroxyproline is a major component of collagen, comprising around 13.5% of its amino acid composition. The basis of hydroxyproline quantification is that total collagen can be assessed by acid hydralization of proteins followed by measurement of hydroxyproline content.
The hydroxyproline content of mouse lung was determined by standard methods. The pulmonary vasculature was perfused with PBS until free of blood. Lung tissue was excised, trimmed free of surrounding conducting airways, and homogenized in 1.8 ml 0.5 mol/L glacial acetic acid. This mixture was dried in a speed vacuum and weighed. The dried sample was redissolved sample in 2 ml of 6N HCl and hydrolyzed at high temperature overnight. 7μl samples were transferred to Eppendorf tubes, dried, and resuspended in citrate-acetate buffer (5% citric acid, 1.2% glacial acetic acid, 7.24% sodium acetate, and 3.4% NaOH dissolved in distilled water and adjusted to pH 6.0). Freshly prepared chloramine-T solution [0.282 g Chloramine-T (Merck), 2 ml n-propanol, 2 ml distilled H2O to 20 ml with citrate-acetate buffer], 150 μl, was added and the sample allowed to stand at room temperature for 20 minutes. 150 μl freshly prepared Ehrlich’s solution [4.5 g 4-dimethylaminobenzaldehyde (Merck) dissolved in 18.6 ml n-propanol, 7.8 ml of 70% perchloric acid] was added and the sample heated to 65°C for 15 minutes. Optical density was measured at 550nm. A standard curve of samples with known quantity of hydroxyproline (Sigma) was generated for each assay. Absorbance of unknown samples was compared to the standard curve and hydroxyproline content per lung was calculated.
Cytokine analysis of Balf fluid and peripheral blood
The BD CBA Mouse Th1/Th2 Ctyokine Kit (Catalog NO 551287) was used to measure Interleukin 2, (IL-2), Interleukin4 (IL-4), Interleukin5 (IL-5), Interferon-γ (IFN- γ), Tumour necrosis factor (TNF) protein levels in both Balf fluid and Peripheral blood samples. This uses bead array technology to simultaneously detect multiple cytokines in the samples. 5 bead populations with distinct fluorescence intensities are coated with capture antibodies, specific for the above mentioned proteins. The beads are mixed to form the bead array, and resolved in a red channel of a flow cytometer. After addition of the sample to the sample assay tubes containing the capture beads, the mouse Th1/Th2 PE detection reagents was added to each tubes. The tubes were incubated for 1 hour at room temperature, in the dark, and then washed with 1ml of wash buffer, centrifuge at 200g for 5 minutes. The supernatant was carefully discarded and 300μl of wash buffer added to resuspended the bead pellet.
Statistical method
Results are given as means ± SD. The differences were assessed by 1 tail Student’s t-test, one-way at α=5% using Graph Pad Prism 6. A P value of <0.05 was considered to indicate a significant difference. Blue star denotes significance in samples compare to control, red star denotes significance in samples treated with only bleo, yellow star denotes significance in samples treated with fisetin, curcumin, and MCN treated fisetin compared to bleo
Results
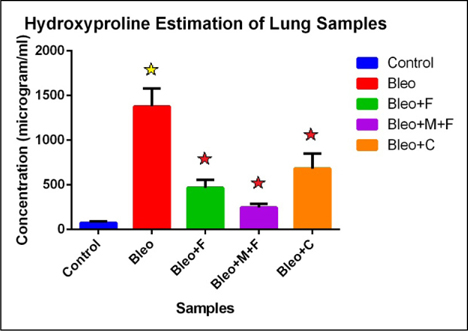
Figure 1 showing comparison between control and treated sample with respect of their hydroxyproline concentration. We found that there is 19 fold increase in collagen concentration for bleo treated group, 6 fold increase for bleo+fisetin treated group , 3 fold increase for bleo+fisetin+ MCN treated group and 9 fold increase for bleo+curcumin treated group. Here we observed that bleomycin is highly effective in causing fibrosis in mice as compared to control groups and the palnts flavonoids also showed their antifibrotic action.
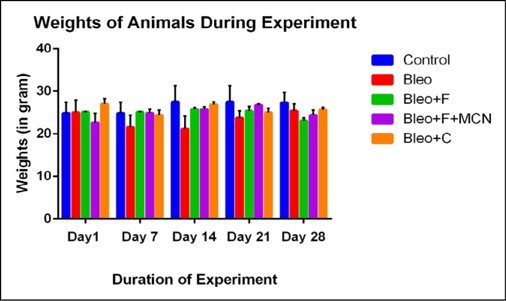
The average weights of different groups of mice were significantly different at the 0thday. During the treatment mainly from the day 7 ( Figure 2 ) we observed that the weight of bleo treated groups were significantly reduced. Weight reduced by 45%, 42% and 19 % on 7th ,14th and 21st day respectively as compared with control groups. When we challenged the bleo treated group with flavonoids (curcumin and fisetin) the weight of the mice were significantly increased 39%, 37% and 5% at day 7,14 and 21strespectively for the curcumin treated group; 55%, 33% and 9% at the respective days for fisetin treated groups and 46%, 33% and 14% at the respective days for MCN loaded fisetin groups. Significant weight loss indicates development of fibrosis after treating with bleomycin.
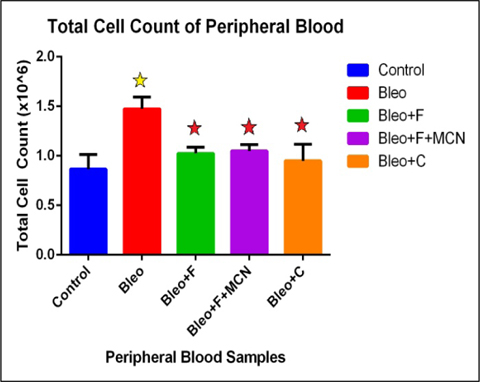
We found that, total cell count in peripheral circulating blood increases 1.7 fold in the bleo treated group as compared to control. We also found that, the count decreases 1.43 fold with fisetin, 1.40 fold with MCN+fisetin, and 1.5 fold with curcumin when compared to only bleo. This indicates 1.3 fold increase with fisetin, and 1.2 fold increase with MCN+fisetin, and 1 fold increase as compared to control treated groups ( Figure 3 ). These findings indicate that after the development of fibrosis during bleomycin treatment the total cell count was greatly increased which was decreased significantly after challenging with the plant flavonoids.
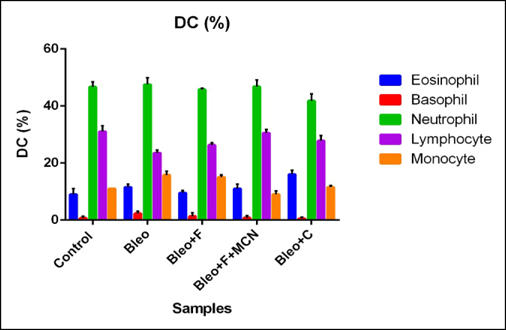
The polymorphonuclear (PMN) cell count was increased by 5.6 % and the mononuclear (MN) cell count by 2% for bleo treated as compared to placebo groups. Both PMN and MN cell counts decreased after treatment with fisetin, curcumin and MCN loaded fisetin. PMN cell count decreased 5% by fisetin treatment and 4% by both curcumin and MCN loaded fisetin treatment, and the MN cell count decreased by 3% for fisetin and 5% by both curcumin and MCN loaded fisetin as compared to only bleo treated samples. Here we found that the inflammatory cell count was greatly increased for bleo treated individuals and effectiveness of fisein was increased after addition of MCN with it, curcumin also showed anti-inflammatory effects ( Figure 4 ).
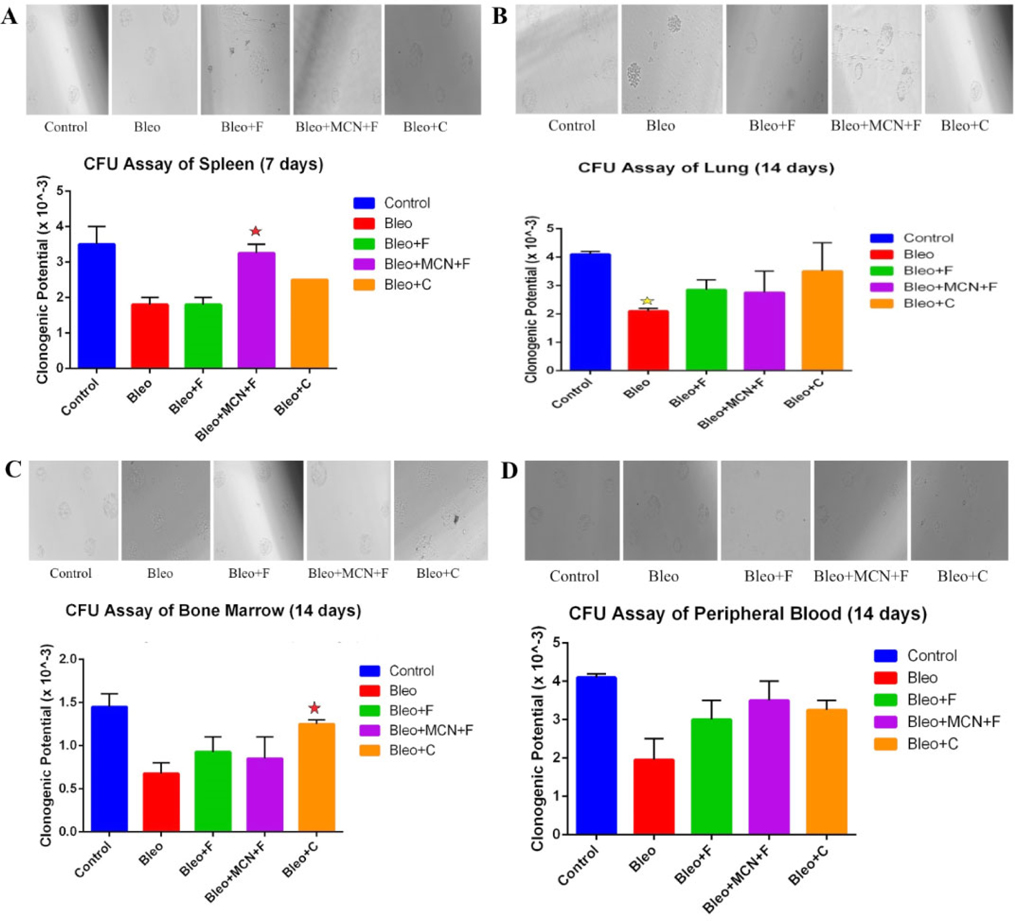
The clonogenic potential in spleen decreases 1.94 fold in the bleo treated as compared to control. The count decreases 1.94 fold with fisetin, 1.07 fold with MCN+fisetin, and 1.4 fold with curcumin as compared to control. 1.80 fold increase with MCN+fisetin, and 1.38 fold increase with curcumin as compared to only bleo. Bleomycin effectively inhibits the cellular recruitment to the spleen and treatment with fisetin, and curcumin increase the cellular recruitment in spleen. Colony count was also increased in MCN+F treated groups, and it shows a statistical significance as compared both with bleo and bleo + fisetin ( Figure 5a ).
The clonogenic potential in lung decreases 2 fold significantly (p < 0.05) than the untreated control groups after challenge with bleomycin, whereas the total number of lung progenitor cell increase 1.35 fold with fisetin 1.30 fold with MCN loaded fisetin and 1.66 fold with curcumin as compared to the bleo treated groups. This denotes that bleomycin effectively decreasing the clonogenic potential of the lung cells and fisetin, MCN loaded fisetin and curcumin also shows its regenerative effect on it among them curcumin showed the best results ( Figure 5b ).
The clonogenic potential of bone marrow (aspirated from the femur) the site of poiesis decreases 2 fold than the untreated control groups after bleomycin challenge whereas in fisetin treated groups, total cell number in bone marrow increases 1.36, in MCN+Fisetin treated groups, total cell number increases 4 fold and for curcumin treated 1.83 fold when compare to only bleo. This indicates a 126.43 fold decrease with Fisetin, and a 2.82 fold decrease with MCN+Fisetin, as compared to only Ova. This shows that the synthesis of cells in the bone marrow, which decreases after challenge with bleomycin, is increased due to the treatment ( Figure 5c ).
Clonogenic potential of peripheral blood was also decreased (2 fold ) for bleo treated groups, treatment with the plant flavonoids trying to show its regenerative affect against bleomycin, among the 3 groups here MCN loaded fisetin showed the best affect, it increases the clonogenic potential by 2 fold as compared to bleo treated groups, fisetin and curcumin also showed its regenerative affect by increasing the clonogenic potential 1.9 fold and 1.7 fold respectively compared to bleo treated groups ( Figure 5d ).
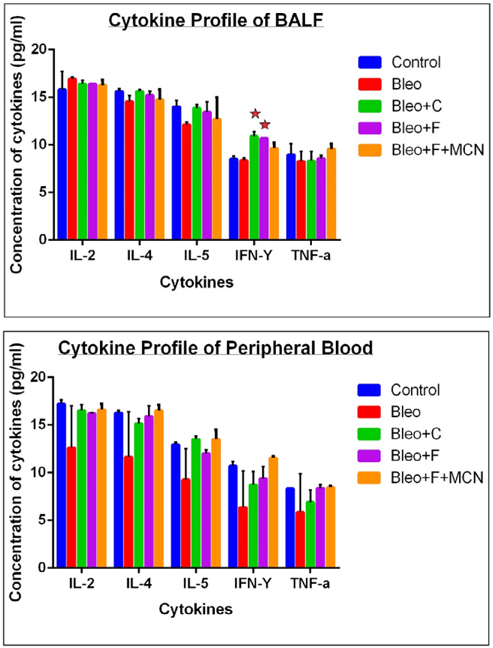
Figure 6a demonstrated that, after challenge with bleomycin, there was an decrease in the levels of IL-4 (1.06 fold), IL-5 (1.16 fold), IFN-γ (1.01 fold) and TNF- α (1.08 fold) as compared to control except IL-2 (1.06 fold increase), there was an increase in the level of cytokines with fisetin treatment (1.03 fold for IL-2, 1.04 fold for IL-4, 1.11 fold for IL-5, 1.11 fold for IFN-γ and1.03 fold for TNF-α),with curcumin treatment (1.03 fold for IL-2, 1.06 fold for IL-4, 1.14 fold for IL-5, 1.29 fold for IFN-γ and1 fold for TNF-α) and with MCN +F treatment (1.03 fold for IL-2, 1.01 fold for IL-4, 1.04 fold for IL-5, 1.15 fold for IFN-γ and1.15 fold for TNF- α) as compared to the bleo treated samples.
After challenge with bleomycin, there was an decrease in the levels of IL-2 (1.36 fold), IL-4 (1.39 fold), IL-5 (1.04 fold), IFN-γ (1.67 fold) and TNF-α (1.41 fold) as compared to control , there is an increase in the level of cytokines with curcumin treatment (1.31 fold for IL- 2, 1.30 fold for IL-4, 1.45 fold for IL-5, 1.35 fold for IFN-γ and1.17 fold for TNF-α) ,with fisetin treatment (1.29 fold for IL-2, 1.35 fold for IL-4, 1.29 fold for IL-5, 1.46 fold for IFN-γ and1.42 fold for TNF-α) and with MCN +fisetin treatment (1.38 fold for IL-2, 1.41 fold for IL-4, 1.45 fold for IL-5, 1.81 fold for IFN-γ and1.44 fold for TNF-α) as compared to the bleo treated sample ( Figure 6b ).
Discussion
For the first time, our study presents evidence that fisetin, curcumin and mesoporous carbaon nanoparticle (MCN) loaded fisetin could be a promising therapeutic option against idiopathic pulmonary fibrosis (IPF).
Bleomycin-induced pulmonary fibrosis is a well established disease model for IPF and widely used in the investigation of the efficacy and mechanism of therapeutic candidates Song et al., 2010. IPF is a progressive interstitial pneumonia of unknown etiology, while its pathogenesis is not fully understood Raghu, 2011. The risk of developing IPF is likely due to both host and environmental factors, and their elucidation may lead to improved prevention and treatment strategies Ley and Collard,2013. Elevated levels of TNF-α, a cytokine with inflammatory and fibrogenic properties have been detected in the lungs in patients with IPF. IPF remains a major cause of morbidity and mortality around the world, and there is still a large unmet medical need. There is no definitive approach to the treatment of IPF because evidence for effective medical therapy is still lacking. A better understanding of the basic biology of IPF is crucial to enable the development of novel therapeutic agents for this disease. Animal models supported the concept not only of abnormal vascular remodelling as a pathogenic mechanism in pulmonary fibrosis (PF), with reports of newly formed vascular networks within the fibrotic lung, but also of increased capillary irregularity and dilatation Peao et al., 1994. Progressive fibrosis with loss of normal lung tissue leads to restricted ventilation, impaired gas exchange, respiratory symptoms and exercise limitation, poor quality of life, and ultimately death Ley and Collard, 2013. Bleomycin is a chemotherapeutic drug used clinically for a variety of human malignancies, including lymphoma. It has been reported that administration of a high dose of bleomycin often leads to lethal lung injury and pulmonary fibrosis in human patients, as well as in rodent Hoshino et al., 2009. Therefore, it has been widely used in making pulmonary fibrosis animal models Lee et al., 2014. The bleomycin model has the advantage that it is quite easy to perform, widely accessible and reproducible, and therefore fulfills important criteria expected from a good animal model Mouratis and Aidinis, 2011. Bleomycin inhibits the incorporation of thymidine, causing DNA fragmentation resulting in apoptosis with the release of chemical mediators for recruitment of immune cells. On the other hand, the epidermal atrophy could be related to the effect of bleomycin, which has been shown to cause cessation of the epidermal cell cycle, with induction of epidermal cell apoptosis Juniantito et al., 2013Kandeel and Balaha, 2015. The bleomycin model has contributed tremendously to elucidate the roles of cytokines, growth factors and signaling pathways involved in pulmonary fibrosis. For instance, it has helped to determine transforming growth factor (TGF) β as one of the key factors in the development of pulmonary fibrosis Zhao et al., 2002.
Unfortunately, no pharmacologic therapy exists at this time that has been proven to improve survival. Due to the heterogeneity of IPF’s clinical course, lack of complete understanding of the pathogenesis and infrequency of the disease itself, there is a lack of largescale randomized controlled trials. During the last decade, several clinical trials for IPF have been conducted worldwide to determine an effective treatment regimen for IPF, but the results have been disappointing. Clinical trial failures may arise for many reasons, including disease heterogeneity, lack of readily measurable clinical end points other than overall survival, and, perhaps most of all, a lack of understanding of the underlying molecular mechanisms of the progression of IPF (Camelo et al., 2014). Some clinical trials are presently assessing the utility of novel agents in the treatment of IPF. Most medications are not recommended due to their side effects and lack of proven benefit Kekevian et al., 2014.
Fisetin has recently received attention for its beneficial effects against several diseases. In the past years, fisetin was a subject of research because of its antiproliferative Haddad et al., 2006Khan et al., 2008Suh et al., 2009, apoptotic Suh et al., 2009Sung et al., 2007, and antioxidant Hou et al., 2001 activities. Several studies indicate that fisetin is a promising novel antioxidant. Fisetin has been reported to inhibit human low-density lipoprotein (LDL) oxidation in vitro Myara et al., 1993. It induced quinone oxidoreductase activity in murine hepatoma 1c1c7 cells in a time- and dose-dependent manner, and the induction of activity was associated with increase in mRNA expression. It was found that fisetin prevented LDL from oxidation, in part, through reducing CD36 gene expression in macrophages, a possible effect in ameliorating atherosclerosis Lian et al., 2008. Curcumin is the active ingredient in the traditional herbal remedy and dietary spice turmeric. Research has revealed that curcumin has a surprisingly wide range of beneficial properties, including antiinflammatory, antioxidant, chemopreventive and chemotherapeutic activity. These activities have been demonstrated both in cultured cells and in animal models, and have paved the way for ongoing human clinical trials Hatcher et al., 2008. Previous report showed that curcumin inhibits the production of proinflammatory monocyte/macrophage-derived cytokines in PMA- or LPS-stimulated peripheral blood monocytes and alveolar macrophages (Abe et al., 1999). Clinical trials of curcumin in humans have been promising. Phase I studies demonstrated virtually no toxicity in humans consuming up to 8g curcumin per day for 3 months or a single dose of up to 12g Hsu et al., 2002Lao et al., 2006. Based on the encouraging preclinical and phase I clinical data, several additional human trials have been initiated and are currently enrolling patients. This includes trials testing the activity of curcumin in patients with colon cancer, pancreatic cancer, multiple myeloma, and myelodisplasia Jiao et al., 2009. Nanoparticles have promising applications in medicine Jiao et al., 2014. Recently, targeted and triggered drug delivery systems accompanied by nanoparticle technology have emerged as prominent solutions to the bioavailability of therapeutic agents. The factors affecting the immune response are complex, including particle composition, size, surface chemistry, plasma protein binding, and exposure route. Investigation of the relationship between properties of nanoparticles and systemic immune response is crucial for their application in medicine and other areas.
In our study, we found that bleomycin is highly effective in causing fibrosis in mice as compare to control groups and the plants flavonoids also showed their anti fibrotic action ( Figure 1 ). The average weights of different groups of mice were significantly different at the 0th day, and the weight of bleo treated groups were significantly reduced mainly from the day 7 ( Figure 2 ). We demonstrated that, total cell count in peripheral circulating blood increases 1.7 fold in the bleo treated group as compared to control. The count decreases 1.43 fold with fisetin, 1.40 fold with MCN+fisetin, and 1.5 fold with curcumin as compared to only bleo. This indicates 1.3 fold increase with fisetin, and 1.2 fold increase with MCN+fisetin, and 1 fold increase as compared to control treated groups ( Figure 3 ).These indicates that after the development of fibrosis during bleomycin treatment the total cell count was greatly increased which was decreased significantly after challenging with the plant flavonoids. In this study, the inflammatory cell count was greatly increased for bleo treated individuals and effectiveness of fisein was increased after addition of MCN particles with it, curcumin also showed anti- inflammatory effects ( Figure 4 ). In another experiment, bleomycin effectively inhibits the cellular recruitment to the spleen and treatment with fisetin, and curcumin increases the cellular recruitment in spleen. Colony count was also increased in MCN+fisetin treated groups, and it was statistically significant ( Figure 5a ). Figure 5b demonstrated that, blebleomycin effectively decreasing the clonogenic potential of the lung cells and fisetin, MCN loaded fisetin and curcumin also showed its regenerative effect on it; among them curcumin showed the best results. We also observed the increased level of cytokines with fisetin treatment, with curcumin treatment and with MCN +fisetin treatment as compared to the bleo treated sample ( Figure 6a ). In other experimental assays, fisetin, curcumin and MCN+fisetin showed promising results in bleomycin-induced idiopathic pulmonary fibrosis in mice.
It is important to emphasize that probably no single agent is going to work in this disease and that a combination, including agents fisetin and curcumin will be necessary. Improvement of our knowledge about the biopathological principles of the disease will increase the opportunities of finding new agents.
Conclusion
In conclusion, Idiopathic pulmonary fibrosis is a challenging, terminal disease characterized by progressive dyspnea and cough and the present research suggests that fisetin and curcumin may be a promising therapeutic agent for bleomycin-induced changes in mice with IPF. Additionally, the administration of mesoporous carbaon nanoparticle loaded fisetin enhanced the beneficial effects. This may open up new perspectives for a potential role of these drugs as a molecular target in Idiopathic pulmonary fibrosis.
Abbreviations
IPF: Idiopathic pulmonary fibrosis, AEC: alveolar epithelial cell, VEGF: vascular endothelial growth factor, PDGF: platelet derived growth factor, FGF: fibroblast growth factor, ILD: interstitial lung disease, DPLD: diffuse parenchymal lung disease, IIP: idiopathic interstitial pneumonia, BALF: broncho alveolar lavage fluid, MCN: mesoporous carbon nano particle, PB: peripheral blood, BM: bone marrow, TC: total count,DC: differential count, IMDM: iscoves modified dulbecco modified medium, CFU: colony forming unit, BLEO: bleomycin, NIN: national institute of nutrition, RHMP: rodent health monitering programme, COX2: cyclooxygenase 2, AP1: activator protein 1, IL-2: Interleukin 2, IL-4: Interleukin 4, IL-5: Interleukin 5, IFN- γ: Interferon-γ, TNF: Tumour necrosis factor, TGF: transforming growth factor, PMN: polymorphonuclear.
References
-
D.L.
Clarke.
The epithelium in idiopathic pulmonary fibrosis: breaking the barrier. Front Pharmacol.
2014;
4
:
173-173
.
-
N.I.
Chaudhary,
G.J.
Roth,
F.
Hilberg,
J.
Muller-Quernheim,
A.
Prasse,
G.
Zissel,
A.
Schnapp,
J.E.
Park.
Inhibition of PDGF, VEGF and FGF signalling attenuates fibrosis. Eur Respir J.
2007;
29
:
976-985
.
-
L.-J.
Chen,
H.
Ye,
Q.
Zhang,
F.-Z.
Li,
L.-J.
Song,
J.
Yang,
Q.
Mu,
S.-S.
Rao,
P.-C.
Cai,
F.
Xiang.
Bleomycin induced epithelial-mesenchymal transition (EMT) in pleural mesothelial cells. Toxicol Appl Pharmacol.
2015;
283
:
75-82
.
-
S.E.
Chuang,
M.L.
Kuo,
C.H.
Hsu,
C.R.
Chen,
J.K.
Lin,
G.M.
Lai,
C.Y.
Hsieh,
A.L.
Cheng.
Curcumin-containing diet inhibits diethylnitrosamine-induced murine hepatocarcinogenesis. Carcinogenesis.
2000;
21
:
331-335
.
-
H.R.
Collard,
T.E.
King,
B.B.
Bartelson,
J.S.
Vourlekis,
M.I.
Schwarz,
K.K.
Brown.
Changes in clinical and physiologic variables predict survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med.
2003;
168
:
538-542
.
-
V.
Cottin,
J.-F.
Cordier.
Velcro crackles: the key for early diagnosis of idiopathic pulmonary fibrosis?. Eur Respir J.
2012;
40
:
519-521
.
-
V.
Cottin,
B.
Crestani,
D.
Valeyre,
B.
Wallaert,
J.
Cadranel,
J.C.
Dalphin,
P.
Delaval,
D.
Israel-Biet,
R.
Kessler,
M.
Reynaud-Gaubert.
Diagnosis and management of idiopathic pulmonary fibrosis: French practical guidelines. European respiratory review : an official journal of the European Respiratory Society.
2014;
23
:
193-214
.
-
W.R.
Coward,
G.
Saini,
G.
Jenkins.
The pathogenesis of idiopathic pulmonary fibrosis. Ther Adv Respir Dis.
2010;
4
:
367-388
.
-
D.
Deeb,
H.
Jiang,
X.
Gao,
S.
Al-Holou,
A.L.
Danyluk,
S.A.
Dulchavsky,
S.C.
Gautam.
Curcumin [1,7-bis(4-hydroxy-3-methoxyphenyl)-1-6-heptadine-3,5-dione; C21H20O6] sensitizes human prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L-induced apoptosis by suppressing nuclear factor-kappaB via inhibition of the prosurvival Akt signaling pathway. J Pharmacol Exp Ther.
2007;
321
:
616-625
.
-
A.
Francois,
A.
Gombault,
B.
Villeret,
G.
Alsaleh,
M.
Fanny,
P.
Gasse,
S.M.
Adam,
B.
Crestani,
J.
Sibilia,
P.
Schneider.
B cell activating factor is central to bleomycin- and IL-17-mediated experimental pulmonary fibrosis. Journal of autoimmunity.
2015;
56
:
1-11
.
-
P.
Gasse,
C.
Mary,
I.
Guenon,
N.
Noulin,
S.
Charron,
S.
Schnyder-Candrian,
B.
Schnyder,
S.
Akira,
V.F.
Quesniaux,
V.
Lagente.
IL-1R1/MyD88 signaling and the inflammasome are essential in pulmonary inflammation and fibrosis in mice. The Journal of clinical investigation.
2007;
117
:
3786-3799
.
-
A.
Goel,
A.B.
Kunnumakkara,
B.B.
Aggarwal.
Curcumin as “Curecumin”: from kitchen to clinic. Biochemical pharmacology.
2008;
75
:
787-809
.
-
J.
Gribbin,
R.B.
Hubbard,
I.
Le Jeune,
C.J.P.
Smith,
J.
West,
L.J.
Tata.
Incidence and mortality of idiopathic pulmonary fibrosis and sarcoidosis in the UK. Thorax.
2006;
61
:
980-985
.
-
S.C.
Gupta,
A.K.
Tyagi,
P.
Deshmukh-Taskar,
M.
Hinojosa,
S.
Prasad,
B.B.
Aggarwal.
Downregulation of tumor necrosis factor and other proinflammatory biomarkers by polyphenols. Arch Biochem Biophys.
2014;
559
:
91-99
.
-
A.Q.
Haddad,
V.
Venkateswaran,
L.
Viswanathan,
S.J.
Teahan,
N.E.
Fleshner,
L.H.
Klotz.
Novel antiproliferative flavonoids induce cell cycle arrest in human prostate cancer cell lines. Prostate Cancer Prostatic Dis.
2006;
9
:
68-76
.
-
H.
Hatcher,
R.
Planalp,
J.
Cho,
F.M.
Torti,
S.V.
Torti.
Curcumin: from ancient medicine to current clinical trials. Cellular and molecular life sciences : CMLS.
2008;
65
:
1631-1652
.
-
T.
Hoshino,
M.
Okamoto,
Y.
Sakazaki,
S.
Kato,
H.A.
Young,
H.
Aizawa.
Role of proinflammatory cytokines IL-18 and IL-1beta in bleomycin-induced lung injury in humans and mice. Am J Respir Cell Mol Biol.
2009;
41
:
661-670
.
-
D.X.
Hou,
M.
Fukuda,
J.A.
Johnson,
K.
Miyamori,
M.
Ushikai,
M.
Fujii.
Fisetin induces transcription of NADPH:quinone oxidoreductase gene through an antioxidant responsive element-involved activation. Int J Oncol.
2001;
18
:
1175-1179
.
-
C.H.
Hsu,
S.E.
Chuang,
M.
Hergenhahn,
M.L.
Kuo,
J.K.
Lin,
C.Y.
Hsieh,
A.L.
Cheng.
Pre-clinical and early-phase clinical studies of curcumin as chemopreventive agent for endemic cancers in Taiwan. Gan to kagaku ryoho Cancer & chemotherapy 29 Suppl.
2002;
1
:
194-200
.
-
Q.
Jiao,
L.
Li,
Q.
Mu,
Q.
Zhang.
Immunomodulation of nanoparticles in nanomedicine applications. Biomed Res Int.
2014;
2014
:
426028-426028
.
-
Y.
Jiao,
J.t.
Wilkinson,
X.
Di,
W.
Wang,
H.
Hatcher,
N.D.
Kock,
R. Jr.
D’Agostino,
M.A.
Knovich,
F.M.
Torti,
S.V.
Torti.
Curcumin, a cancer chemopreventive and chemotherapeutic agent, is a biologically active iron chelator. Blood.
2009;
113
:
462-469
.
-
C.
Jobin,
C.A.
Bradham,
M.P.
Russo,
B.
Juma,
A.S.
Narula,
D.A.
Brenner,
R.B.
Sartor.
Curcumin blocks cytokine-mediated Nfkappa B activation and proinflammatory gene expression by inhibiting inhibitory factor I-kappa B kinase activity. Journal of immunology.
1999;
(Baltimore
:
Md : 1950) 163, 3474-3483
.
-
V.
Juniantito,
T.
Izawa,
T.
Yuasa,
C.
Ichikawa,
M.
Tanaka,
M.
Kuwamura,
J.
Yamate.
Immunophenotypical analysis of myofibroblasts and mesenchymal cells in the bleomycin-induced rat scleroderma, with particular reference to their origin. Exp Toxicol Pathol.
2013;
65
:
567-577
.
-
S.
Kandeel,
M.
Balaha.
The possible protective effect of simvastatin and pioglitazone separately and in combination on bleomycininduced changes in mice thin skin. Tissue Cell.
2015
.
-
A.
Kekevian,
M.E.
Gershwin,
C.
Chang.
Diagnosis and classification of idiopathic pulmonary fibrosis. Autoimmun Rev.
2014;
13
:
508-512
.
-
N.
Khan,
M.
Asim,
F.
Afaq,
M.
Abu Zaid,
H.
Mukhtar.
A novel dietary flavonoid fisetin inhibits androgen receptor signaling and tumor growth in athymic nude mice. Cancer Res.
2008;
68
:
8555-8563
.
-
T.E.
King,
J.
Behr,
K.K.
Brown,
R.M.
du Bois,
L.
Lancaster,
J.A.
de Andrade,
G.
Stahler,
I.
Leconte,
S.
Roux,
G.
Raghu.
BUILD-1: a randomized placebo-controlled trial of bosentan in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med.
2008;
177
:
75-81
.
-
T.E.
King,
A.
Pardo,
M.
Selman.
Idiopathic pulmonary fibrosis. Lancet.
2011;
378
:
1949-1961
.
-
Y.
Kondoh,
H.
Taniguchi,
Y.
Kawabata,
T.
Yokoi,
K.
Suzuki,
K.
Takagi.
Acute exacerbation in idiopathic pulmonary fibrosis. Analysis of clinical and pathologic findings in three cases.. Chest.
1993;
103
:
1808-1812
.
-
C.D.
Lao,
M.T.t.
Ruffin,
D.
Normolle,
D.D.
Heath,
S.I.
Murray,
J.M.
Bailey,
M.E.
Boggs,
J.
Crowell,
C.L.
Rock,
D.E.
Brenner.
Dose escalation of a curcuminoid formulation. BMC complementary and alternative medicine.
2006;
6
:
1-0
.
-
R.
Lee,
C.
Reese,
M.
Bonner,
E.
Tourkina,
Z.
Hajdu,
E.C.
Riemer,
R.M.
Silver,
R.P.
Visconti,
S.
Hoffman.
Bleomycin delivery by osmotic minipump: similarity to human scleroderma interstitial lung disease. Am J Physiol Lung Cell Mol Physiol.
2014;
306
:
736-748
.
-
B.
Ley,
H.R.
Collard.
Epidemiology of idiopathic pulmonary fibrosis. Clin Epidemiol.
2013;
5
:
483-492
.
-
T.-W.
Lian,
L.
Wang,
Y.-H.
Lo,
I.J.
Huang,
M.-J.
Wu.
Fisetin, morin and myricetin attenuate CD36 expression and oxLDL uptake in U937-derived macrophages. Biochim Biophys Acta.
2008;
1781
:
601-609
.
-
E.D.
Manali,
C.
Moschos,
C.
Triantafillidou,
A.
Kotanidou,
I.
Psallidas,
S.P.
Karabela,
C.
Roussos,
S.
Papiris,
A.
Armaganidis,
G.T.
Stathopoulos.
Static and dynamic mechanics of the murine lung after intratracheal bleomycin. BMC Pulm Med.
2011;
11
:
33-33
.
-
Y.
Manolova,
V.
Deneva,
L.
Antonov,
E.
Drakalska,
D.
Momekova,
N.
Lambov.
The effect of the water on the curcumin tautomerism: a quantitative approach. Spectrochim Acta A Mol Biomol Spectrosc.
2014;
132
:
815-820
.
-
A.
Moeller,
K.
Ask,
D.
Warburton,
J.
Gauldie,
M.
Kolb.
The bleomycin animal model: a useful tool to investigate treatment options for idiopathic pulmonary fibrosis?. Int J Biochem Cell Biol.
2008;
40
:
362-382
.
-
M.A.
Mouratis,
V.
Aidinis.
Modeling pulmonary fibrosis with bleomycin. Curr Opin Pulm Med.
2011;
17
:
355-361
.
-
I.
Myara,
I.
Pico,
B.
Vedie,
N.
Moatti.
A method to screen for the antioxidant effect of compounds on low-density lipoprotein (LDL): illustration with flavonoids. J Pharmacol Toxicol Methods.
1993;
30
:
69-73
.
-
M.N.
Peao,
A.P.
Aguas,
C.M.
de Sa,
N.R.
Grande.
Neoformation of blood vessels in association with rat lung fibrosis induced by bleomycin. Anat Rec.
1994;
238
:
57-67
.
-
S.
Quideau,
D.
Deffieux,
C.
Douat-Casassus,
L.
Pouysegu.
Plant polyphenols: chemical properties, biological activities, and synthesis. Angewandte Chemie (International ed in English).
2011;
50
:
586-621
.
-
G.
Raghu.
Idiopathic pulmonary fibrosis: guidelines for diagnosis and clinical management have advanced from consensus-based in 2000 to evidence-based in 2011. Eur Respir J.
2011;
37
:
743-746
.
-
G.
Raghu,
H.R.
Collard,
J.J.
Egan,
F.J.
Martinez,
J.
Behr,
K.K.
Brown,
T.V.
Colby,
J.-F.
Cordier,
K.R.
Flaherty,
J.A.
Lasky.
An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med.
2011a;
183
:
788-824
.
-
G.
Raghu,
H.R.
Collard,
J.J.
Egan,
F.J.
Martinez,
J.
Behr,
K.K.
Brown,
T.V.
Colby,
J.F.
Cordier,
K.R.
Flaherty,
J.A.
Lasky.
An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med.
2011b;
183
:
788-824
.
-
G.
Raghu,
T.D.
Freudenberger,
S.
Yang,
J.R.
Curtis,
C.
Spada,
J.
Hayes,
J.K.
Sillery,
C.E.
Pope,
C.A.
Pellegrini.
High prevalence of abnormal acid gastro-oesophageal reflux in idiopathic pulmonary fibrosis. Eur Respir J.
2006;
27
:
136-142
.
-
B.D.
Sahu,
A.K.
Kalvala,
M.
Koneru,
J.
Mahesh Kumar,
M.
Kuncha,
S.S.
Rachamalla,
R.
Sistla.
Ameliorative effect of fisetin on cisplatin-induced nephrotoxicity in rats via modulation of NF-κB activation and antioxidant defence. PLoS One.
2014;
9
.
-
M.
Selman,
T.E.
King,
A.
Pardo.
Idiopathic pulmonary fibrosis: prevailing and evolving hypotheses about its pathogenesis and implications for therapy. Annals of internal medicine.
2001;
134
:
136-151
.
-
J.S.
Song,
C.M.
Kang,
H.H.
Kang,
H.K.
Yoon,
Y.K.
Kim,
K.H.
Kim,
H.S.
Moon,
S.H.
Park.
Inhibitory effect of CXC chemokine receptor 4 antagonist AMD3100 on bleomycin induced murine pulmonary fibrosis. Exp Mol Med.
2010;
42
:
465-472
.
-
Y.
Suh,
F.
Afaq,
J.J.
Johnson,
H.
Mukhtar.
A plant flavonoid fisetin induces apoptosis in colon cancer cells by inhibition of COX2 and Wnt/EGFR/NF-kappaB-signaling pathways. Carcinogenesis.
2009;
30
:
300-307
.
-
B.
Sung,
M.K.
Pandey,
B.B.
Aggarwal.
Fisetin, an inhibitor of cyclin-dependent kinase 6, down-regulates nuclear factor-kappaBregulated cell proliferation, antiapoptotic and metastatic gene products through the suppression of TAK-1 and receptor-interacting proteinregulated IkappaBalpha kinase activation. Mol Pharmacol.
2007;
71
:
1703-1714
.
-
H.
Taniguchi,
M.
Ebina,
Y.
Kondoh,
T.
Ogura,
A.
Azuma,
M.
Suga,
Y.
Taguchi,
H.
Takahashi,
K.
Nakata,
A.
Sato.
Pirfenidone in idiopathic pulmonary fibrosis. Eur Respir J.
2010;
35
:
821-829
.
-
L.
Wollin,
I.
Maillet,
V.
Quesniaux,
A.
Holweg,
B.
Ryffel.
Antifibrotic and anti-inflammatory activity of the tyrosine kinase inhibitor nintedanib in experimental models of lung fibrosis. J Pharmacol Exp Ther.
2014;
349
:
209-220
.
-
J.H.
Woo,
Y.H.
Kim,
Y.J.
Choi,
D.G.
Kim,
K.S.
Lee,
J.H.
Bae,
D.S.
Min,
J.S.
Chang,
Y.J.
Jeong,
Y.H.
Lee.
Molecular mechanisms of curcumin-induced cytotoxicity: induction of apoptosis through generation of reactive oxygen species, down-regulation of Bcl-XL and IAP, the release of cytochrome c and inhibition of Akt. Carcinogenesis.
2003;
24
:
1199-1208
.
-
C.
Yamazaki,
J.
Hoshino,
T.
Sekiguchi,
Y.
Hori,
S.
Miyauchi,
S.
Mizuno,
K.
Horie.
Production of superoxide and nitric oxide by alveolar macrophages in the bleomycin-induced interstitial pneumonia mice model. Japanese journal of pharmacology.
1998;
78
:
69-73
.
-
Z.
Yan,
Z.
Kui,
Z.
Ping.
Reviews and prospectives of signaling pathway analysis in idiopathic pulmonary fibrosis. Autoimmun Rev.
2014;
13
:
1020-1025
.
-
J.
Zhao,
W.
Shi,
Y.L.
Wang,
H.
Chen,
P. Jr.
Bringas,
M.B.
Datto,
J.P.
Frederick,
X.F.
Wang,
D.
Warburton.
Smad3 deficiency attenuates bleomycin-induced pulmonary fibrosis in mice. Am J Physiol Lung Cell Mol Physiol.
2002;
282
:
L585-593
.
Comments

Downloads
Article Details
Volume & Issue : Vol 2 No 04 (2015)
Page No.: 250-262
Published on: 2015-04-30
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
- HTML viewed - 6825 times
- Download PDF downloaded - 2036 times
- View Article downloaded - 8 times
 Biomedpress
Biomedpress