Abstract
Background: The nootropic or simply known as smart drug is a common term given to any compound that is responsible for enhancing mental capability or performance. Alzheimer's disease is characterized clinically by lose of cognitive abilities and pathologically by two hallmark lesions, neurofibrillary tangles and senile plaques. It is unfortunate that AD has no cure yet. In this review attempt has been made to elucidate the general views on AD pathogenic hypotheses and common nootropics being used in AD research.
Methods: Articles from credible scientific data bases such as Sciencdirect, Scopus Pubmed, and Google scholar were searched and retrieved using keywords nootropics', Alzheimer's disease', amyloid beta hypotheses', tau hypotheses', cholinergic hypotheses', oxidative stress' and cognitive impairments'.
Results: The nootropics act as Ca-channel blockers, AChE inhibitors, glysine antagonists, antioxidants, serotonergic, dopaminergic and glutamic acid receptors antagonists.
Conclusion: Based on the available literature searched, there is no doubts the nootropics are attenuating cognitive deficits in both preclinical and clinical studies on AD.
Introduction
Alzheimer’s disease (AD) is the most common type of age-related dementia, characterized by a progressive decline in hippocampal-dependent functions, including cognitive deterioration, memory loss, behavioral and functional disturbances, and functional impairment 1,2. Clinically, AD is diagnosed by dementia, while pathologically it is diagnosed by two cardinal lesions including senile plaques caused by the extracellular deposits of amyloid beta fibrils and neurofibrillary tangles formed by the abnormal intracellular aggregation of tau protein 3. The etiopathogenesis of AD is multifactorial. Oxidative stress and cholinergic dysfunction have been suggested to play a vital role in the onset and progression of the disease 4,5. During the progression of AD, neurons from different parts of the brain are destroyed, including those areas that enable basic bodily functions like swallowing and walking. Hence, AD patients in the final stage usually become bedridden and eventually dies 6. Advances in science and technology, as well as good healthcare delivery, have increased life expectancy. Unfortunately, it is accompanied by a cost of a higher frequency of age-related diseases such as AD.
The “nootropic” or simply known as “smart drug”, “memory-enhancing drug” or “brain booster” is a common terminology given to compounds with the ability to enhance mental performance 7, although people with a history of mental disorder may be susceptible to its adverse effects 8. By definition, nootropics are compounds that increase mental abilities including attention, concentration, memory, and motivation 7.
There has been a lot of research conducted on AD’s prevention and treatment strategies. Although different approaches were implemented to lessen the progression of the disease, there is no cure for AD. Hence, nootropics have been explored for this purpose and have been yielding some promising results. This review aimed to elucidate the general view on AD pathogenic hypotheses and common nootropics being used in AD research. All the vital information required for this review was gathered by searching the relevant keywords including, nootropics, Alzheimer’s disease, amyloid beta hypotheses, tau hypotheses, cholinergic hypotheses, oxidative stress, and cognitive impairments from published articles. The search on nootropics is basically from 2011 to 2018, and reliable scientific databases were searched namely ScienceDirect, Scopus, PubMed, and Google Scholar.
Alzheimer’s disease pathogenesis: general views
AD is a heterogeneous disorder with divergent clinical symptomatology, various ages of onset, presence or absence of germline mutations, degree and spread of pathological changes, existence or non-existence of risk factors and manifestation or non-appearance of polymorphic susceptibility alleles. Therefore, it is not surprising that several hypotheses with “treatment insinuations” have been proposed. However, it is unfortunate that none of these hypotheses have led to tangible treatment benefits or a cure for AD 9. The most widely acclaimed hypotheses in the scientific community include amyloid cascade hypotheses, tau hypotheses, cholinergic hypotheses, and oxidative stress among others.
Amyloid cascade hypothesis
The vast majority of research in the field of AD have focused on amyloid cascade hypotheses since the early 90s when AD research began to gain momentum. The amyloid cascade hypothesis states that “the pathogenic cascade of AD initiates upon accumulation, oligomerization, and aggregation of the amyloid beta peptide (Aβ) in extracellular deposits termed senile plaques” 10. This aggregation of senile plaques consequently stimulate the hyperphosphorylation of tau protein, leading to the formation of neurofibrillary tangles and neurodegeneration. The creeds of the amyloid beta hypotheses are generally founded on the existence of rare autosomal, early-onset forms of AD, all of which involved mutations that affect the processing of APP and resulted into increased production and accumulation of Aβ. Based on these explanations, the hypothesis assumes that reversing, halting or preventing this process will cure the disease 10. This current evidence based on amyloid cascade hypotheses does not support the pathogenesis of sporadic form of AD which is 95% of AD cases diagnosed 11. Therefore, this view envisages that all amyloid-beta-centered therapies for AD will continue to be unsuccessful 12. It is also unfortunate that developing alternative therapies could not be possible unless the etiology of AD is well understood.
Tau hypothesis
As senile plaque and neuronal loss does not completely correlate in AD, research have turned towards other characteristics well known to AD, backing-up the idea known as the “tau hypothesis”13. The tau hypothesis of the pathogenesis of AD suggests that tau abnormality facilitates neurotoxicity and neurodegeneration and these are important contributors to the development of AD 14. Tau is a microtubule-associated protein (MAP) with amino acids ranging from 352-441 in length. Six Tau isoforms in the adult human brain are all derived from a single gene called MAPT gene through alternative RNA splicing. The primary role of tau is to regulate the stability of microtubules. Another physiological function of tau is to allow signaling molecules, neurotransmitters, and trophic factors to travel along axons 15. Under normal physiological conditions, tau is in a continuous dynamic equilibrium of short biding and detachment to microtubules through phosphorylation by kinases and dephosphorylation by phosphatases respectively. These cycles ensue an effective axonal transport 16. Phosphorylation and dephosphorylation of tau happen under regular physiological conditions, with supposed hyperphosphorylation happening in diseased brains 17. The imbalance of phosphorylation causes phosphorylated tau to detach from microtubules and aggregate into dense paired-helical filaments within the cell, which eventually kills the neuron 13. Antibodies were developed to remove toxic tau with the hope that by doing so, it will stop or slow the progression of AD 18. However, none of the antibodies has been shown to be effective. Though the impact of tau in distracting the functions of neurons is clear, it has been shown that tau aggregation occurs at the late stage of AD pathogenesis where intervention is likely ineffective. For tau hypotheses to be accepted, there must be evidence that tau dysregulation is both the main initiator and occurs very early in the disease onset 12.
The Cholinergic hypotheses of AD
The cholinergic hypothesis was the pioneer theory suggested to explain the etiology of AD and had led to the development of the only drug that is approved by US food and drugs authority for the treatment of mild to moderate AD 19,20. The theory was based on the fact that a loss of cholinergic activity is regularly observed in AD patients brains 21. And, results from multiple studies in human and animal have suggested a role of acetylcholine in cognitive functions. These studies reported that blocking the central cholinergic activity with scopolamine could induce memory deficits in young subjects to behave like old individuals. On the other hand, administration of cholinergic agonist physostigmine could reverse the cognitive impairment 22. Based on the cholinergic theory, another type of cholinergic agonist, acetylcholinesterase inhibitors (AChEIs) were developed and have shown effectiveness in reversing cognitive impairments in AD patients. Small improvements in cognitive abilities have been reported in some clinical trials with AChEIs as compared to placebo. However, the effects are not permanent as patients showed cognitive deterioration over time 23,24. In addition, some AD patients are not responding to AChEIs treatments, and the difference between responders and non-responders has not been discovered 25,26. Others have reported a decrease in acetylcholine (ACh) level does not cause severe memory impairments in rats 27,28. Some researchers have used this finding together with the failure of AChEIs to cure AD as a reason to invalidate the cholinergic hypotheses and shifted their attention towards mutations of APP genes, tau protein production, and Aβ depositions as the more likely causative factors 29,30.
Notwithstanding, other researchers continued to explore the role of ACh in the development of AD with the hopes that one day they could explain the dysfunction of ACh in AD brains. Craig proposed a modified cholinergic hypothesis by suggesting that the depletion of the neurotransmitter ACh reduces the ability of the brain to compensate for secondary insults that come with the aging process 31. With the plethora of AD risk factors, there are many questions needed to be addressed relating to this hypothesis such as if aging the only risk factor to be considered, how the modified cholinergic hypothesis explains the early onset form of AD, and what causes of the ACh depletion? These questions will open discussion for more specific causative factors and theories.
Oxidative stress hypotheses of AD
Recent studies have developed a keen interest in the role of oxidative stress in neurologic disorders. There are indications that free radicals have a role in Parkinson’s disease (PD), Down’s syndrome (DS), head injury, cerebral ischemia-reperfusion, and AD 32. The CNS is particularly sensitive to damages induced by free radical because of the high lipid content, high oxygen utilization rate, and less presence of antioxidant enzymes in the brain compared to other tissues 33. The most interesting part of the oxidative stress hypothesis for neurodegenerative diseases is that accumulative oxidative injury over a long time could lead to a late onset and progression of neurodegenerative disease 33,34,35.
Studies on transgenic animal models, biological fluids from DS, mild cognitive impairment (MCI) and AD patients, cell culture models, and postmortem brains have demonstrated the involvement of oxidative stress in the early stage of these disorders. Apart from an increase of several oxidative stress markers in AD, there is also evidence of lower antioxidant power in the blood, CSF, and brain of AD patients 36. GSH is the most predominant antioxidant in brain cells. GSH can react with oxidized products and ROS to create glutathione disulfide (GSSG), either catalyzed independently or by Glutathione Peroxidase (GPx). The GSSG can be further reconverted back to GSH by Glutathione Reductase (GR). Studies of lymphocytes from AD patients have shown that the ratio between reduced and oxidized glutathione (GSH/GSSG) is decreased 37, this is also observed in the brain of AD patients 38, and in the hippocampus of MCI patients 39.
In spite of several studies indicating the strong effects of oxidative stress in the pathogenesis of neurodegenerative diseases and the use of antioxidants in reduction or prevention of damage caused by free radicals, the efficacy in clinical trials is still controversial. Results from clinical trials using antioxidants for prevention or treatment of AD have been so far disappointing, and there are several reasons for these failures 36. The possible reasons for the failed clinical trials with antioxidants could be; (i) small sample size enrolled for the trials which are not good enough for statistical analysis; (ii) short-term duration of antioxidant interventions which could not give enough time for the desired outcomes to surface; (iii) dosages used during the trials as high dose may produce adverse effect while low dose could not be enough to produce good results; (iv) poor choice of antioxidant for specific effects in most cases several antioxidants are needed instead of one; (v) clinical conditions of the AD patients enrolled for the trials since there are multiple signaling pathways that can generate oxidative stress 36,40. The arguments are still open-ended; should antioxidant therapy be continued or discontinued, what are the other possible options for preventing, slowing or stopping AD progression and which of the hypotheses should be adopted for more effective treatments?
Nootropics
Nootropics, also known as “smart drugs”, are compounds that have been developed over the past 35 years and perhaps the first to be used for the treatment of cognitive deficits 41. The word nootropic coin from Greek word (“noos” means “to mind” and “tropein” means “to monitor”) is used to define in a wide range, any substance that is accredited with the ability to enhance cognition and support healthy brain function 42. The nootropics can be broadly classified into two categories: the naturally occurring, such as Centella asiatica, Ginkgo biloba, and Panax quinquefolius among others and synthetic nootropic, a laboratory created compounds such as Piracetam, modafinil, and racetams 41. These types of substances include a number of agents like cholinergic, serotonergic, dopaminergic, and antioxidants drugs. However, for this work, we analyze nootropics that are specifically used to target AD and few brain injuries, the reason for this choice is to shed light on these promising agents in the fight against AD. Below are highlights of some studies on nootropics and the major findings, both animal and human studies were reviewed. To gain more insight into the pharmacodynamics of nootropics, recent studies on cell lines as well as review papers were also considered.
Dichrocephala integrifolia improved cognitive deficits and attenuated neuronal death on scopolamine-induced mouse model of AD 43, while Pharmaceutical substance (PhS) based on amide form Human leukemia differential factor -6 (AF HLDF-6) restores cognitive dysfunction in C57B1/6 transgenic mouse model of AD 44. Further, fisetin reverses synaptic dysfunction, prevented neuro-inflammation, and improves memory in C57BL/6N transgenic mouse model of cognitive dysfunction 45, and Cerebrolysin showed its neuroprotective potential by protecting grafted Neural stem cells (NSCs) in an APP Transgenic mice model of AD, hence it could be a potential adjuvant therapy for AD when combined with grafting 46. Treatment of rat model of AD with “2-(2-benzofuranyl)-2-imidazoline (2-BFI) restored cognitive impairments, attenuated oxidative stress, and protects against inflammation and apoptosis in a dose-dependent manner 47. Simvastatin ameliorated cognitive impairment and inflammation in both rat model of AD and clinical patients of AD by modulating the expression of MicroRNA106b (miR-106b) as reported by Huang 48. Similarly, Centella asiatica (CA) an Ayurvedic herb attenuated cognitive impairments in d-galactose and aluminum chloride induced rats through the prevention of apoptosis and ultrastructural alterations of hippocampal neurons 49. CA was also reported to attenuate Aβ induced oxidative stress and mitochondrial dysfunction in vitro 50,51 and improve spatial memory in animals 52. Increased in synaptic density and improvement in executive functions was also observed in healthy aged mice after treatment with CA 53. Similarly, short treatment with CA has increased the expression of synaptic, mitochondrial, and antioxidant response genes and improved different domains of cognitive performance (executive function, memory, and learning) in 5xFAD animals as well as reduced the burden of Aβ plaque burden in the hippocampus 54.
Lipopolysaccharide (LPS) induced oxidative stress, cell death and inflammatory response in Sprague Dawley rats were reversed by piracetam through attenuation against mitochondria-mediated caspase-independent pathway 55. A new Tacrine-Hydroxyphenylbenzimidazole (TAC-BIM) hybrid compound with excellent multifunctional activity was developed and tested on Human neuroblastoma SH-SY5Y Cell lines. The chemical inhibits AChE activity better than drug tacrine by preventing self-induced or Copper-induced Aβ aggregation, having antioxidant activity, and showing neuroprotective capacity against Aβ 56. Another nootropic compound “L-theamine” protects SH-SY5Y cells against glutamate-induced toxicity through inhibition of N-methyl-D-aspartate (NMDA) glutamate subtype of receptors and related pathways. Hence, L-theamine may serve as prophylaxis and treatment for AD 57. Further, Piracetam exerts its neuroprotective effects on PC12 cells, SH-SY5Y cells, and SH-SY5Y APPwt cells by improving synaptic plasticity, maintaining mitochondrial dynamic and neuritogenesis 58. In addition, Noopept protects PC12 cells against Aβ25-35 induced toxicity by inhibiting of oxidative damage, preventing of calcium overload, suppressing apoptosis, attenuating hyperphosphorylation of tau, and ameliorating the neural outgrowth induced by Aβ25-35 59.
In addition to the preclinical studies, several clinical trials were also conducted on nootropics in AD patients and related dementias with impressive results. Galantamine was tested on 50 aggressive AD patients and had a significant improvement in Zarit burden interview (ZBI) scores after 12 weeks of treatment 60. Similarly, a historical cohort study was conducted on 33 patients with severe disability after traumatic brain injury (TBI), administration of cerebrolysin decreased mortality rate and improved the functional recovery in TBI patients though it had seizure as an adverse sideeffect 61. In another development, huperzine A and curcumin were given as supplements to AD, MCI, and other dementia patients. The patients showed improvements in their cognitive functions as measured by AD assessment scale-cognitive subscale Japanese version (ADAS-Jcog) 62. Conversely, in three randomized clinical trials on 2525 AD patients addition of idalopirdine as an adjunct to cholinesterase inhibitors did not improve cognitive deficits over a period of 24 weeks 63. Further, a retrospective observational study was conducted on 189 AD patients, no significant difference in cognitive decline was observed between donepezil and Ginkgo biloba extract in over 12 months as measured with Mini mental state examination (MMSE) score although more adverse side effects were seen in donepezil 64. Another retrospective study was conducted on 2570 AD patients. The study suggested that using of statins might be beneficial to all AD patients especially those with homozygous for Apolipoprotein E4 (ApoE4) 65.
Because of the numerous preclinical and clinical studies on nootropics, several reviews were also conducted to harness the impressive outcomes. The neurocognitive effects of brahmi on cognitive impaired experimental animals was reviewed, the authors opined that brahmi could be a good candidate against AD in human patients 66. Similarly, the use of galantamie, donepezil, and rivastigmine in mild to moderate AD provide modest cognitive function and behavior as reviewed by Mohammad 67. Furthermore, profiling donepezil template into multipotent hybrids through molecular docking has shown that it has anticholinesterase activity with escalating antioxidant potential 68.
Conclusion
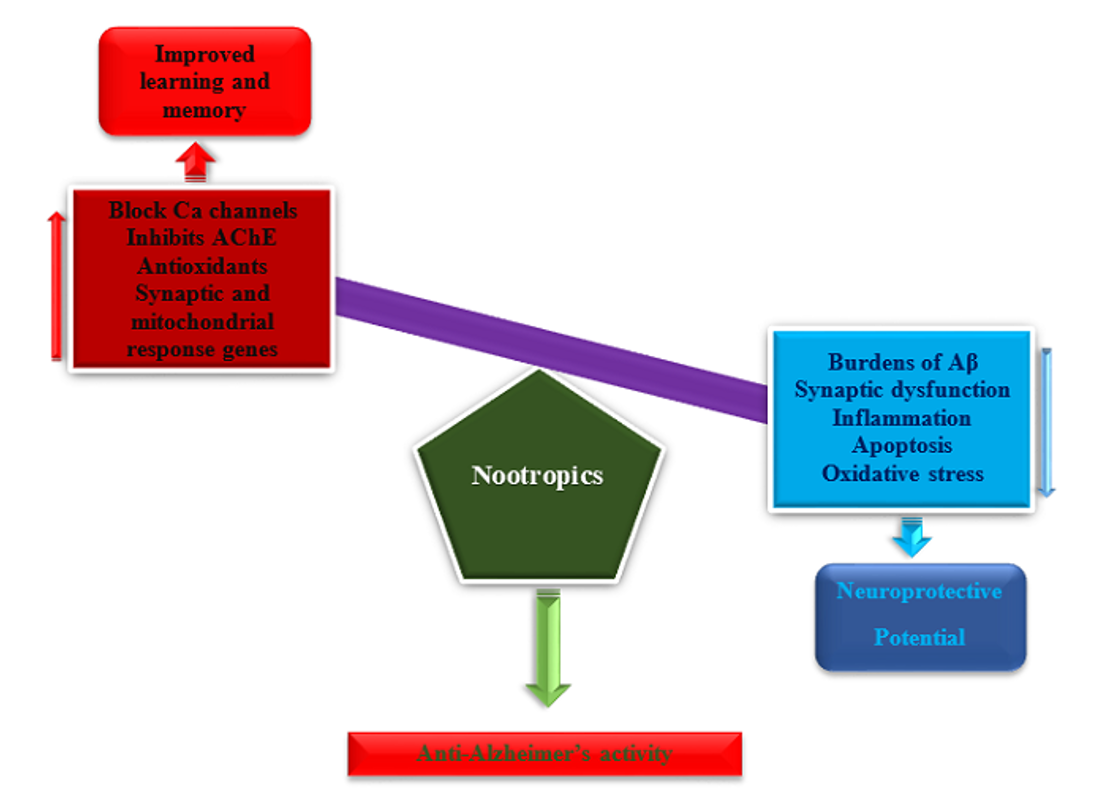
Scientists in the field of AD research has been working vigorously on nootropics, which has expanded the understanding of the mechanism of action of both synthetic and natural nootropics for the past 35 years. The nootropics improve memory and learning by acting as Ca-channel blockers, AChEI, glycine antagonists, antioxidants, serotonergic, dopaminergic, and glutamic acid receptors antagonists. Further, nootropics exhibit neuroprotective potentials by decreasing the burden of Aβ accumulation, apoptosis, synaptic dysfunction, inflammation and oxidative stress (Figure 1). Based on the available literature searched, both on pre-clinical and clinical effects of the nootropics in AD, there is no doubt some of the obtained results are encouraging. Some animal AD models and cell lines responded well to the treatments with nootropics, which was further studied in AD patients. However, clinical trials with a small number of patients cannot serve as a basis for a meaningful assessment of clinical efficacy. In addition, the data obtained on the efficacy or inefficacy of the nootropics could be impractical because of the intrinsic problems with clinical trials. For example, can patients who responded well to Ca-channel blockers do the same to AChE inhibitors or what pathological or physiological difference that separates responders to non-responders? Therefore, there is still a need to study further on nootropics with multimodal targets, with the hope it could finally bring light at the end of the tunnel.
Abbreviations
2-BFI: 2-(2-benzofuranyl)-2-imidazoline
AChE: Acetylcholinesterase
AD: Alzheimer’s disease
ADAS-Jcog: AD assessment scale cognitive sub-scale Japanese version
ApoE4: Apolipoprotein E4
Aβ: Beta amyloid
Cu: Copper
HLDF-6: Human leukemia differential factor -6
LPS: Lipopolysacchride
MCI: Mild cognitive impairment
miR-106b: MicroRNA106b
MMSE: Mini mental state examination
NMDA: N-methyl-D-aspartate
NSCs: Neural stem cells
PhS: Pharmaceutical substance:
TAC-BIM: Tacrine-Hydroxyphenylbenzimidazole
TBI: Traumatic brain injury
ZBLPS: Zarit caregiver burden interview
Competing Interests
The authors declare that they have no conflicts of interest.
Authors' Contributions
All authors contributed to the design of the research. MSC and SJ extracted the data and summarized it. MTBH, CNMT, ZA and MAMM edited the first draft. All authors reviewed, commented and approved the final draft.
Acknowledgments
The authors would like to acknowledge Universiti Putra Malaysia for funding this research project (Grant number GP-IPS 9535400).
References
-
Cooper
E. L.,
Ma
M. J..
Alzheimer Disease: clues from traditional and complementary medicine. Journal of Traditional and Complementary Medicine.
2017;
7
:
380-5
.
View Article PubMed Google Scholar -
Deshmukh
R.,
Sharma
V.,
Mehan
S.,
Sharma
N.,
Bedi
K. L..
Amelioration of intracerebroventricular streptozotocin induced cognitive dysfunction and oxidative stress by vinpocetine-- a PDE1 inhibitor. European Journal of Pharmacology.
2009;
620
:
49-56
.
View Article PubMed Google Scholar -
Firuzi
O.,
Pratico
D..
Coxibs and Alzheimer's disease: should they stay or should they go?. Annals of Neurology.
2006;
59
:
219-28
.
View Article PubMed Google Scholar -
Weinreb
O.,
Amit
T.,
Bar-Am
O.,
Youdim
M. B..
Ladostigil: a novel multimodal neuroprotective drug with cholinesterase and brain-selective monoamine oxidase inhibitory activities for Alzheimer\'s disease treatment. Current Drug Targets.
2012;
13
:
483-94
.
View Article Google Scholar -
Santos
D. B.,
Peres
K. C.,
Ribeiro
R. P.,
Colle
D.,
Santos
A. A. dos,
Moreira
E. L..
Probucol, a lipid-lowering drug, prevents cognitive and hippocampal synaptic impairments induced by amyloid ? peptide in mice. Experimental Neurology.
2012;
233
:
767-75
.
View Article PubMed Google Scholar -
Association
A..
2018 Alzheimer''s disease facts and figures. Alzheimer\'s & Dementia.
2018;
14
:
367-429
.
View Article Google Scholar -
Lanni
C.,
Lenzken
S. C.,
Pascale
A.,
Vecchio
I. Del,
Racchi
M.,
Pistoia
F..
Cognition enhancers between treating and doping the mind. Pharmacological Research.
2008;
57
:
196-213
.
View Article PubMed Google Scholar -
Garcia
H. Saiz,
Reula
L. Montes,
Fernandez
A. Portilla,
Sanchez
V. Pereira,
Lopez
N. Olmo,
Heredero
E. Mancha.
Nootropics: emergents drugs associated with new clinical challenges. European Psychiatry.
2017;
41
:
S877-8
.
View Article Google Scholar -
Castellani
R. J.,
Zhu
X.,
Lee
H. G.,
Smith
M. A.,
Perry
G..
Molecular pathogenesis of Alzheimer's disease: reductionist versus expansionist approaches. International Journal of Molecular Sciences.
2009;
10
:
1386-406
.
View Article PubMed Google Scholar -
Karran
E.,
Mercken
M.,
Strooper
B. De.
The amyloid cascade hypothesis for Alzheimer's disease: an appraisal for the development of therapeutics. Nature Reviews. Drug Discovery.
2011;
10
:
698-712
.
View Article PubMed Google Scholar -
Thonberg
H.,
Chiang
H. H.,
Lilius
L.,
Forsell
C.,
Lindstrom
A. K.,
Johansson
C..
Identification and description of three families with familial Alzheimer disease that segregate variants in the SORL1 gene. Acta Neuropathologica Communications.
2017;
5
:
43
.
View Article PubMed Google Scholar -
Castello
M. A.,
Soriano
S..
On the origin of Alzheimer's disease. Trials and tribulations of the amyloid hypothesis. Ageing Research Reviews.
2014;
13
:
10-2
.
View Article PubMed Google Scholar -
Iqbal
K.,
Liu
F.,
Gong
C. X.,
Alonso
A. C.,
Grundke-Iqbal
I..
Mechanisms of tau-induced neurodegeneration. Acta Neuropathologica.
2009;
118
:
53-69
.
View Article Google Scholar -
Desai
A. K.,
Chand
P..
Tau-based Therapies for Alzheimer' s Disease : wave of the Future?. Primary Psychiatry.
2009;
16
:
40-6
.
-
Dixit
R.,
Ross
J. L.,
Goldman
Y. E.,
Holzbaur
E. L..
Differential regulation of dynein and kinesin motor proteins by tau. Science.
2008;
319
:
1086-9
.
View Article PubMed Google Scholar -
Kosik
K. S..
Traveling the tau pathway: a personal account. Journal of Alzheimer's Disease.
2006;
9
:
251-6
.
View Article PubMed Google Scholar -
Lace
G. L.,
Wharton
S. B.,
Ince
P. G..
A brief history of tau: the evolving view of the microtubule-associated protein tau in neurodegenerative diseases. Clinical Neuropathology.
2007;
26
:
43-58
.
View Article Google Scholar -
Yanamandra
K.,
Kfoury
N.,
Jiang
H.,
Mahan
T. E.,
Ma
S.,
Maloney
S. E..
Anti-tau antibodies that block tau aggregate seeding in vitro markedly decrease pathology and improve cognition in vivo. Neuron.
2013;
80
:
402-14
.
View Article PubMed Google Scholar -
Bartus
R. T.,
Dean
R. L.,
Beer
B.,
Lippa
A. S..
The cholinergic hypothesis of geriatric memory dysfunction. Science.
1982;
217
:
408-14
.
View Article Google Scholar -
Bartus
R. T..
On neurodegenerative diseases, models, and treatment strategies: lessons learned and lessons forgotten a generation following the cholinergic hypothesis. Experimental Neurology.
2000;
163
:
495-529
.
View Article PubMed Google Scholar -
Davies
P.,
Maloney
A. J..
Selective loss of central cholinergic neurons in Alzheimer's disease. Lancet.
1976;
2
:
1403
.
View Article PubMed Google Scholar -
Bartus
R. T..
Evidence for a direct cholinergic involvement in the scopolamine-induced amnesia in monkeys: effects of concurrent administration of physostigmine and methylphenidate with scopolamine. Pharmacology, Biochemistry, and Behavior.
1978;
9
:
833-6
.
View Article Google Scholar -
Doody
R. S.,
Dunn
J. K.,
Clark
C. M.,
Farlow
M.,
Foster
N. L.,
Liao
T..
Chronic donepezil treatment is associated with slowed cognitive decline in Alzheimer\'s disease. Dementia and Geriatric Cognitive Disorders.
2001;
12
:
295-300
.
View Article PubMed Google Scholar -
Courtney
C.,
Farrell
D.,
Gray
R.,
Hills
R.,
Lynch
L.,
Sellwood
E.,
null
null.
Long-term donepezil treatment in 565 patients with Alzheimer\'s disease (AD2000): randomised double-blind trial. Lancet.
2004;
363
:
2105-15
.
View Article Google Scholar -
Connelly
P. J.,
Prentice
N. P.,
Fowler
K. G..
Predicting the outcome of cholinesterase inhibitor treatment in Alzheimer\'s disease. Journal of Neurology, Neurosurgery, and Psychiatry.
2005;
76
:
320-4
.
View Article PubMed Google Scholar -
Lemstra
A. W.,
Richard
E.,
Gool
W. A. van.
Cholinesterase inhibitors in dementia: yes, no, or maybe?. Age and Ageing.
2007;
36
:
625-7
.
View Article PubMed Google Scholar -
Parent
M. B.,
Baxter
M. G..
Septohippocampal acetylcholine: involved in but not necessary for learning and memory?. Learning & Memory (Cold Spring Harbor, N.Y.).
2004;
11
:
9-20
.
View Article PubMed Google Scholar -
Mesulam
M..
The cholinergic lesion of Alzheimer's disease: pivotal factor or side show?. Learning & Memory (Cold Spring Harbor, N.Y.).
2004;
11
:
43-9
.
View Article PubMed Google Scholar -
Bartus
R. T..
On neurodegenerative diseases, models, and treatment strategies: lessons learned and lessons forgotten a generation following the cholinergic hypothesis. Experimental Neurology.
2000;
163
:
495-529
.
View Article PubMed Google Scholar -
Mesulam
M. M..
Neuroplasticity failure in Alzheimer's disease: bridging the gap between plaques and tangles. Neuron.
1999;
24
:
521-9
.
View Article Google Scholar -
Craig
L. A.,
Hong
N. S.,
McDonald
R. J..
Revisiting the cholinergic hypothesis in the development of Alzheimer\'s disease. Neuroscience and Biobehavioral Reviews.
2011;
35
:
1397-409
.
View Article PubMed Google Scholar -
Markesbery
W. R..
Oxidative stress hypothesis in Alzheimer's disease. Free Radic Biol Med.
1997;
23
:
134-47
.
PubMed Google Scholar -
Coyle
J. T.,
Puttfarcken
P..
Oxidative stress, glutamate, and neurodegenerative disorders. Science.
1993;
262
:
689-95
.
View Article Google Scholar -
Dexter
D.,
Carter
C.,
Agid
F.,
Agid
Y.,
Lees
A. J.,
Jenner
P..
Lipid peroxidation as cause of nigral cell death in Parkinson\'s disease. Lancet.
1986;
2
:
639-40
.
View Article Google Scholar -
Olanow
C. W..
A radical hypothesis for neurodegeneration. Trends in Neurosciences.
1993;
16
:
439-44
.
View Article Google Scholar -
Persson
Torbjörn,
Popescu
Bogdan O,
Cedazo-Minguez
Angel.
Oxidative stress in Alzheimer’s disease: why did antioxidant therapy fail?. Oxidative Medicine and Cellular Longevity.
2014;
2014
.
PubMed Google Scholar -
Calabrese
V.,
Sultana
R.,
Scapagnini
G.,
Guagliano
E.,
Sapienza
M.,
Bella
R..
Nitrosative stress, cellular stress response, and thiol homeostasis in patients with Alzheimer\'s disease. Antioxidants & Redox Signalling.
2006;
8
:
1975-86
.
View Article PubMed Google Scholar -
Benzi
G.,
Moretti
A..
Age- and peroxidative stress-related modifications of the cerebral enzymatic activities linked to mitochondria and the glutathione system. Free Radical Biology & Medicine.
1995;
19
:
77-101
.
View Article Google Scholar -
Sultana
R.,
Piroddi
M.,
Galli
F.,
Butterfield
D. A..
Protein levels and activity of some antioxidant enzymes in hippocampus of subjects with amnestic mild cognitive impairment. Neurochemical Research.
2008;
33
:
2540-6
.
View Article PubMed Google Scholar -
Feng
Y.,
Wang
X..
Antioxidant therapies for Alzheimer's disease. Oxidative Medicine and Cellular Longevity.
2012;
2012
:
472932
.
View Article PubMed Google Scholar -
Suliman
N. A.,
Taib
C. N. Mat,
Moklas
M. A. Mohd,
Adenan
M. I.,
Baharuldin
M. T. Hidayat,
Basir
R..
Establishing Natural Nootropics: Recent Molecular Enhancement Influenced by Natural Nootropic. Evidence-Based Complementary and Alternative Medicine.
2016;
2016
:
4391375
.
View Article PubMed Google Scholar -
Malykh
A. G.,
Sadaie
M. R..
Piracetam and piracetam-like drugs: from basic science to novel clinical applications to CNS disorders. Drugs.
2010;
70
:
287-312
.
View Article PubMed Google Scholar -
Kouemou
N. E.,
Taiwe
G. S.,
Moto
F. C.,
Pale
S.,
Ngoupaye
G. T.,
Njapdounke
J. S..
Nootropic and neuroprotective effects of Dichrocephala integrifolia on scopolamine mouse model of Alzheimer\'s disease. Frontiers in Pharmacology.
2017;
8
:
847
.
View Article PubMed Google Scholar -
Bogachouk
AP,
Storozheva
ZI,
Telegin
GB,
Chernov
AS,
Proshin
AT,
Sherstnev
VV,
Lipkin
VM.
Studying the Specific Activity of the Amide Form of HLDF-6 Peptide using the Transgenic Model of Alzheimer’s Disease. Acta Naturae (англоязычная версия).
2017;
9
.
PubMed Google Scholar -
Ahmad
A.,
Ali
T.,
Park
H. Y.,
Badshah
H.,
Rehman
S. U.,
Kim
M. O..
Neuroprotective Effect of Fisetin Against Amyloid-Beta-Induced Cognitive/Synaptic Dysfunction, Neuroinflammation, and Neurodegeneration in Adult Mice. Molecular Neurobiology.
2017;
54
:
2269-85
.
View Article PubMed Google Scholar -
Rockenstein
E.,
Desplats
P.,
Ubhi
K.,
Mante
M.,
Florio
J.,
Adame
A..
Neuro-peptide treatment with Cerebrolysin improves the survival of neural stem cell grafts in an APP transgenic model of Alzheimer disease. Stem Cell Research (Amsterdam).
2015;
15
:
54-67
.
View Article PubMed Google Scholar -
Tian
J. S.,
Zhai
Q. J.,
Zhao
Y.,
Chen
R.,
Zhao
L. D..
2-(2-benzofuranyl)-2-imidazoline (2-BFI) improved the impairments in AD rat models by inhibiting oxidative stress, inflammation and apoptosis. Journal of Integrative Neuroscience.
2017;
16
:
385-400
.
View Article PubMed Google Scholar -
Huang
W.,
Li
Z.,
Zhao
L.,
Zhao
W..
Simvastatin ameliorate memory deficits and inflammation in clinical and mouse model of Alzheimer\'s disease via modulating the expression of miR-106b. Biomedicine and Pharmacotherapy.
2017;
92
:
46-57
.
View Article PubMed Google Scholar -
Chiroma
S. M.,
Baharuldin
M. T. Hidayat,
Taib
C. N. Mat,
Amom
Z.,
Jagadeesan
S.,
Adenan
M. I..
Protective effect of Centella asiatica against D-galactose and aluminium chloride induced rats: behavioral and ultrastructural approaches. Biomedicine and Pharmacotherapy.
2019;
109
:
853-64
.
View Article PubMed Google Scholar -
Gray
N. E.,
Sampath
H.,
Zweig
J. A.,
Quinn
J. F.,
Soumyanath
A..
Centella asiatica Attenuates Amyloid-?-Induced Oxidative Stress and Mitochondrial Dysfunction. Journal of Alzheimer\'s Disease.
2015;
45
:
933-46
.
View Article Google Scholar -
Gray
N. E.,
Zweig
J. A.,
Murchison
C.,
Caruso
M.,
Matthews
D. G.,
Kawamoto
C..
Centella asiatica attenuates A?-induced neurodegenerative spine loss and dendritic simplification. Neuroscience Letters.
2017;
646
:
24-9
.
View Article PubMed Google Scholar -
Soumyanath
Amala,
Zhong
Yong-Ping,
Henson
Edward,
Wadsworth
Teri,
Bishop
James,
Gold
Bruce G,
Quinn
Joseph F.
Centella asiatica extract improves behavioral deficits in a mouse model of Alzheimer\'s disease: investigation of a possible mechanism of action. International Journal of Alzheimer’s Disease.
2012;
2012
.
-
Gray
N. E.,
Zweig
J. A.,
Caruso
M.,
Martin
M. D.,
Zhu
J. Y.,
Quinn
J. F..
Centella asiatica increases hippocampal synaptic density and improves memory and executive function in aged mice. Brain and Behavior.
2018;
8
:
e01024
.
View Article Google Scholar -
Gray
N. E.,
Zweig
J. A.,
Caruso
M.,
Zhu
J. Y.,
Wright
K. M.,
Quinn
J. F..
Centella asiatica attenuates hippocampal mitochondrial dysfunction and improves memory and executive function in ?-amyloid overexpressing mice. Molecular and Cellular Neurosciences.
2018;
93
:
1-9
.
View Article Google Scholar -
Verma
Dinesh Kumar,
Gupta
Sonam,
Biswas
Joyshree,
Joshi
Neeraj,
Singh
Abhishek,
Gupta
Parul,
Tiwari
Shubhangini,
Raju
K Sivarama,
Chaturvedi
Swati,
Wahajuddin
M.
New therapeutic activity of metabolic enhancer piracetam in treatment of neurodegenerative disease: Participation of caspase independent death factors, oxidative stress, inflammatory responses and apoptosis. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease.
2018;
1864
:
2078-2096
.
-
Hiremathad
A.,
Keri
R. S.,
Esteves
A. R.,
Cardoso
S. M.,
Chaves
S.,
Santos
M. A..
Novel Tacrine-Hydroxyphenylbenzimidazole hybrids as potential multitarget drug candidates for Alzheimer\'s disease. European Journal of Medicinal Chemistry.
2018;
148
:
255-67
.
View Article PubMed Google Scholar -
Di
X.,
Yan
J.,
Zhao
Y.,
Zhang
J.,
Shi
Z.,
Chang
Y..
L-theanine protects the APP (Swedish mutation) transgenic SH-SY5Y cell against glutamate-induced excitotoxicity via inhibition of the NMDA receptor pathway. Neuroscience.
2010;
168
:
778-86
.
View Article Google Scholar -
Stockburger
C.,
Miano
D.,
Pallas
T.,
Friedland
K.,
Muller
W. E..
Enhanced Neuroplasticity by the Metabolic Enhancer Piracetam Associated with Improved Mitochondrial Dynamics and Altered Permeability Transition Pore Function. Neural Plasticity.
2016;
2016
:
8075903
.
View Article PubMed Google Scholar -
Ostrovskaya
R. U.,
Vakhitova
Y. V.,
Kuzmina
U. S.,
Salimgareeva
M. K.,
Zainullina
L. F.,
Gudasheva
T. A..
Neuroprotective effect of novel cognitive enhancer noopept on AD-related cellular model involves the attenuation of apoptosis and tau hyperphosphorylation. Journal of Biomedical Science.
2014;
21
:
74
.
View Article PubMed Google Scholar -
Nakayama
S.,
Suda
A.,
Nakanishi
A.,
Motoi
Y.,
Hattori
N..
Galantamine Response Associates with Agitation and the Prefrontal Cortex in Patients with Alzheimer\'s Disease. Journal of Alzheimer's Disease.
2017;
57
:
267-73
.
View Article PubMed Google Scholar -
Khalili
H.,
Niakan
A.,
Ghaffarpasand
F..
Effects of cerebrolysin on functional recovery in patients with severe disability after traumatic brain injury: A historical cohort study. Clinical Neurology and Neurosurgery.
2017;
152
:
34-8
.
View Article Google Scholar -
Tabira
T.,
Kawamura
N..
A study of a supplement containing huperzine A and curcumin in dementia patients and individuals with mild cognitive impairment. Journal of Alzheimer\'s Disease.
2018;
63
:
75-8
.
View Article PubMed Google Scholar -
Atri
A.,
Frolich
L.,
Ballard
C.,
Tariot
P. N.,
Molinuevo
J. L.,
Boneva
N..
Effect of idalopirdine as adjunct to cholinesterase inhibitors on change in cognition in patients with alzheimer disease: three randomized clinical trials. Journal of the American Medical Association.
2018;
319
:
130-42
.
View Article PubMed Google Scholar -
Rapp
M.,
Burkart
M.,
Kohlmann
T.,
Bohlken
J..
Similar treatment outcomes with Ginkgo biloba extract EGb 761 and donepezil in Alzheimer\'s dementia in very old age: A retrospective observational study. International Journal of Clinical Pharmacology and Therapeutics.
2018;
56
:
130-3
.
View Article Google Scholar -
Geifman
N.,
Brinton
R. D.,
Kennedy
R. E.,
Schneider
L. S.,
Butte
A. J..
Evidence for benefit of statins to modify cognitive decline and risk in Alzheimer's disease. Alzheimer's Research & Therapy.
2017;
9
:
10
.
View Article PubMed Google Scholar -
Chaudhari
K. S.,
Tiwari
N. R.,
Tiwari
R. R.,
Sharma
R. S..
Neurocognitive effect of nootropic drug Brahmi (Bacopa monnieri) in Alzheimer\'s disease. Annals of Neurosciences.
2017;
24
:
111-22
.
View Article PubMed Google Scholar -
Mohammad
D.,
Chan
P.,
Bradley
J.,
Lanctot
K.,
Herrmann
N..
Acetylcholinesterase inhibitors for treating dementia symptoms - a safety evaluation. Expert Opinion on Drug Safety.
2017;
16
:
1009-19
.
View Article PubMed Google Scholar -
Mezeiova
E.,
Spilovska
K.,
Nepovimova
E.,
Gorecki
L.,
Soukup
O.,
Dolezal
R..
Profiling donepezil template into multipotent hybrids with antioxidant properties. Journal of Enzyme Inhibition and Medicinal Chemistry.
2018;
33
:
583-606
.
View Article PubMed Google Scholar
Comments
Downloads
Article Details
Volume & Issue : Vol 6 No 1 (2019)
Page No.: 2937-2944
Published on: 2019-01-04
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 34080 times
- Download PDF downloaded - 3859 times
- View Article downloaded - 0 times
 Biomedpress
Biomedpress


