
Variations of anterior and posterior division of internal iliac artery: A systematic review and clinical implications
- Department of Anatomy, Faculty of Medicine, Ilam University of Medical Sciences, Ilam, Iran
- Student research committee, Ilam University of Medical Sciences, Ilam, Iran
Abstract
The distribution pattern of internal iliac artery (IIA) implies its bifurcation to two branches, the anterior and the posterior trunks. According to previous research, IIA indicates several anatomical variations. The purpose of this study is to evaluate the types of these variations. The presence of these variations in the arteries is an important topic to urologists, gynecologists, radiologists, and general surgeons because they can be ruptured during surgical procedures in the perineal and pelvis region. Three databases were searched for the period from 1810 to January 2018 and a total of 75 studies were investigated. This study evaluated the branching of the internal iliac artery in several literatures and compared it with Adachi's classification. This finding can lead to improve surgical technique and safety in medical practice.
Introduction
From an anatomic point of view, internal iliac artery (IIA) begins as a terminal branch of the common iliac artery at the level of sacroiliac joint. Each IIA is around 3.5 cm long, descending near the upper border of greater sciatic foramen and divides into anterior and posterior branches. The ureter, uterine tube and ovary are placed on the anterior surface of the IIA. However, the internal iliac vein and lumbosacral trunk pass from the posterior side of IIA. The branches arising from the anterior and posterior trunks of the IIA are described in
Branches of anterior and posterior trunk of the IIA in different sex of male and female
Branches of IIA | Male | Female | |
Anterior trunk of the IIA | Parietal branches | Internal pudendal artery | internal pudendal artery |
| Inferior gluteal artery | inferior gluteal artery | ||
| Obturator artery | Obturator artery | ||
Visceral branches | Umbilical artery | Umbilical artery | |
| Superior vesical artery | superior vesical artery | ||
| Inferior vesical arterymiddle rectal artery | Vaginal artery (replaced by inferior vesical artery)middle rectal artery | ||
| uterine artery (only in women) | |||
Posterior trunk of the IIA | Parietal branches | Superior gluteal artery | Superior gluteal artery |
| lateral sacral arteries | lateral sacral arteries | ||
| Iliolumbar artery | Iliolumbar artery | ||
During development, this artery derives from the proximal part of umbilical artery that is also called hypogastric artery whereas the distal part of umbilical artery obliterated postnatally 2,4. This embryological development leads to several variations in the origin of IIA and its branching pattern into two major trunks 3. Hence, understanding anatomical variation of IIA is essential for surgeons to apply the IIA ligature to prevent hemorrhages following by pelvic surgeries, hysterectomies and orthopedic surgeries related to hip joint. Previous studies show that the level of origin of IIA is variable and dependent on the length of the common iliac artery and the level of division of IIA 5. Typically, the origin of the IIA is between L5 and the upper border of S1 1,2. When we searched for the anomalies declared on the normal pattern, IIA shows several anatomical variations. There have been studies that describe different forms of IIA in a population or individually. However, we attempted to create a proper classification of these variations, since each author described his own findings as an independent classification. Hence, none of the categories represent the observed variations completely. In this study, we collected recent reports to:
The first attempt to classify variant patterns of IIA was performed by Jastschinski that showed four types which he described on an investigation of variant patterns of IIA branches. This classification was based on the four branches that included the inferior gluteal artery, internal pudendal artery, superior gluteal artery and umbilical artery 6.
Materials and Methods
Research method
The report of this review study was based on a systematic review and meta-analysis (PRISMA) (7). PUBMED, ISI web of knowledge and SCOPUS were searched for published studies up to January 2018. Three anatomical textbooks (Gray’s Anatomy, SNELL and Netter) were evaluated in order to find any type of evidence in association with IIA. Several key words were used in our search, including “internal iliac artery”, “hypogastric artery”, “arteria iliaca interna”, “anterior division internal iliac artery” and “posterior division internal iliac artery”. To increase the accuracy of the process, two independent researchers carried out writing the papers and evaluating data. The study was evaluated and selected in three stages. In the first step, citation information and a summary of the evaluated papers were transferred to the Endnote software. Then the titles of the selected articles were reviewed and the articles that were not related to the main topic of the research were excluded. In the second stage, from the abstracts, articles related to the main subject were determined. Finally, the original text of the selected articles was assessed.
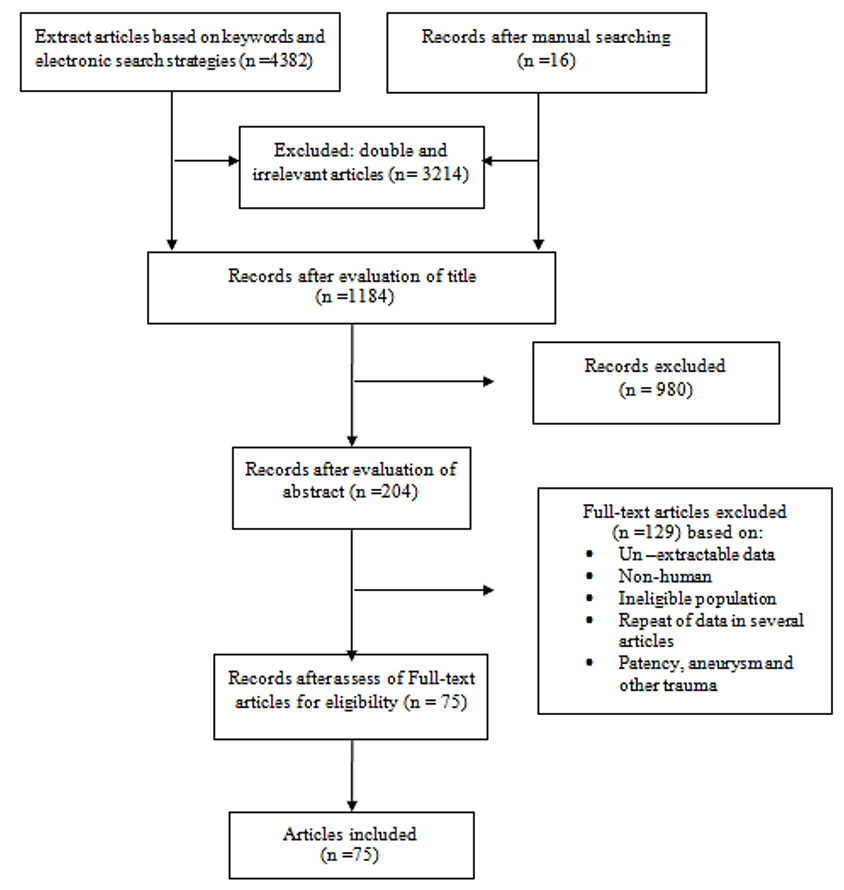
PRISMA diagram of included articles into process.
Inclusion and exclusion criteria
The data included into the study complied with the following criteria:
1) they were consisted of original research, case report and review articles from the anatomical variations of the AII in cadaveric, clinical and imaging sample studies, or those which provided data about variation of the superior vesical artery (SVA), inferior vesical artery (IVA), middle rectal artery (MRA), obturator artery (OA), internal pudendal artery (IPA), inferior gluteal artery (IGA), uterine artery (UtA), vaginal artery (VA), superior gluteal artery (SGA), lateral sacral arteries (LSA) and iliolumbar artery (ILA).
2) Only human studies were selected.
3) There were no restrictions related to the demographic characteristics of the sample study (race, sex, and age).
Study selection and data extraction
The initial electronic and manual search yielded 4398 articles. After extracting all papers, evaluating the titles and removing double and irrelevant articles, 1184 studies were selected. After reviewing the abstracts and their adaptation to the inclusion and exclusion criteria, 204 articles related to the subject matter remained. After evaluating the full texts of the articles, 129 articles were disqualified and 75 articles were confirmed and prepared for final analysis. The process of extracting and selecting articles is shown in Figure 1. The required data included the origin of IIA at different levels, distance from greater sciatic foramen and types of IIA, SVA, IVA, MRA, OA, IPA, IGA, UtA, VA, SGA, LSA, ILA, variations (first author’s name, publication year, sample size and mean).
Results and Discussion
Distribution of the internal iliac artery (IIA)
According to the older terminology, IIA arises from the common iliac artery at the level of the sacroiliac joint and adjusts with the intervertebral disc between L5 and S1, then descends posteriorly within the pelvic cavity; subsequently, it reaches the greater sciatic foramen 1,2,3,4. Several studies reported the variation in the origin of the IIA (
Origin of internal iliac artery (IIA) at different levels
Study | Ref | Vertebral level | No. of specimen | Percentage |
| Mamatha (2015) | L5-S1 vertebraL5 vertebra | 362 | 72*4 | |
| Sakthivelavan (2014) | L5-S1 vertebraAbove level | 9422 | 81*19 | |
| Havaldar (2014) | L5-L4 vertebraS1 vertebra | 82 | 164 | |
| Naveen (2011) | S1 vertebraL5 vertebra | 351 | 58.3*1.7 | |
| Fatu (2006) | Sacro-iliac joint | 100 | 100* |
Distance from greater sciatic foramen
Study | Ref | Distance | No. of specimen | Percentage |
| Mamatha H etal. (2015) | Above 2 cm Below 1 cm | 14 1 | 282 | |
| Havaldar P etal. (2014) | Above 0.5cm below the upper border 2 cm | 92 | 18*4 | |
| Havaldar P etal. (2014) | Above below the upper border | 349 | 68*18 | |
| Sakthivelavan S etal. (2014) | Upper borderposition between lumbosacral articulation and greater sciatic notch | 7940 | 65.5*34.5 | |
| Naveen et al (2011) | Above 3 cmAbove 5.5 cm | 184 | 30.7*6.6 |
The IIA divides at the level of superior edge of greater sciatic notch.
In 1928, this classification was introduced by Adachi in Japanese subjects with a slight change 12. He described IIA variations in five types which include:
Type I: The superior gluteal artery and a common trunk arise from IIA. The common trunk is bifurcate and forms the inferior gluteal and internal pudendal arteries. If the common trunk divides into the pelvis, it is type Ia, and if it is outside the pelvis then it’s classified as type Ib.
Type II: The internal pudendal artery and a common trunk arise from IIA. The common trunk is bifurcate and forms the inferior gluteal and superior gluteal arteries. If the common trunk divides into the pelvis, it is of the type IIa, and if it is outside the pelvis then it is classified as type IIb.
Type III: The internal pudendal artery and two gluteal arteries are given off separately from the internal iliac artery.
Type IV: The internal pudendal artery and two gluteal arteries originate from the same trunk. In type IVa, the trunk first gives rise to the superior gluteal artery while in type IVb, the internal pudendal is the first vessel to be separated.
Type V: The inferior gluteal artery and the common trunk arise from IIA. The common trunk is bifurcate and forms the internal pudendal and superior gluteal arteries into the pelvis (Figure 2).

Adachi’s types of IIA variations. IIA: internal iliac artery; IGA: Inferiorgluteal artery; IPA: Internal pudendal artery; SGA: Superior gluteal artery; UA:Umbilical artery
Types of IIA variations according to Adachi classification (%)
|
Study |
Year | Types of IIA variations according to Adachi classification% | No. of specimen | |||||
I | II | III | IV | V | Other | |||
| Lipshutz | 1916 | 51 | 24 | 17 | 7 | - | - | 181 |
| Adachi | 1928 | 51.2 | 23.1 | 18.2 | 4.1 | 0.8 | 2.5 | 121 |
| Tsukamoto | 1929 | 56.5 | 8.4 | 22 | 12.9 | - | - | 287 |
| Miyaji | 1935 | 70.4 | 11.7 | 9.5 | 8.4 | - | - | 179 |
| Aria | 1936 | 52.4 | 19.4 | 24 | 4.2 | - | - | 500 |
| Hoshiai | 1938 | 55.1 | 16.1 | 26.1 | 2.6 | - | - | 379 |
| Ashley & Anson | 1941 | 58.1 | 17.3 | 9.6 | 7.7 | - | 7.3 | 260 |
| Suzuki | 1951 | 53.2 | 18.8 | 24.1 | 3.7 | 0.2 | - | 490 |
| Braithwaite | 1952 | 58.5 | 15.3 | 22.5 | 3.6 | - | - | 169 |
| Yasukawa | 1954 | 53.7 | 18.4 | 23.9 | 4 | - | - | 544 |
| Fischer | 1959 | 50 | 26 | 16 | 8 | - | - | 50 |
| Roberts & Krishingner | 1968 | 50.9 | 27 | 14.4 | 7.2 | - | 0.6 | 167 |
| Morita | 1974 | 49.1 | 22.5 | 21.7 | 6.7 | - | - | 267 |
| Lwasaki | 1987 | 54.2 | 19.5 | 24.3 | 2 | - | - | 251 |
| Yamaki | 1998 | 58 | 13.6 | 22.8 | 5.4 | 0.2 | - | 645 |
| Sakthivelavan | 2014 | 63.2 | 15.8 | 21 | - | - | - | 116 |
| Talalwah | 2014 | 36.1 | 5.3 | 34.8 | 2.3 | - | - | 342 |
| Present study | 2018 | 54.2 | 17.7 | 20.7 | 5.6 | 0.4 | 3.46 | 4948 |
Anterior trunk of the IIA
Parietal branches
Internal pudendal artery (IPA)
The most common origins of the internal pudendal artery (IPA) are a branch that stems from the anterior division of the IIA and is the main vessel of the perineum. However, several articles confirm the variable source for this artery. It is important for anatomists and surgeons to understand of IPA1,3,4,2. The IPA was classified by 29 in the vicinity of the greater sciatic notch, the linea terminalis, and the ischial spine (
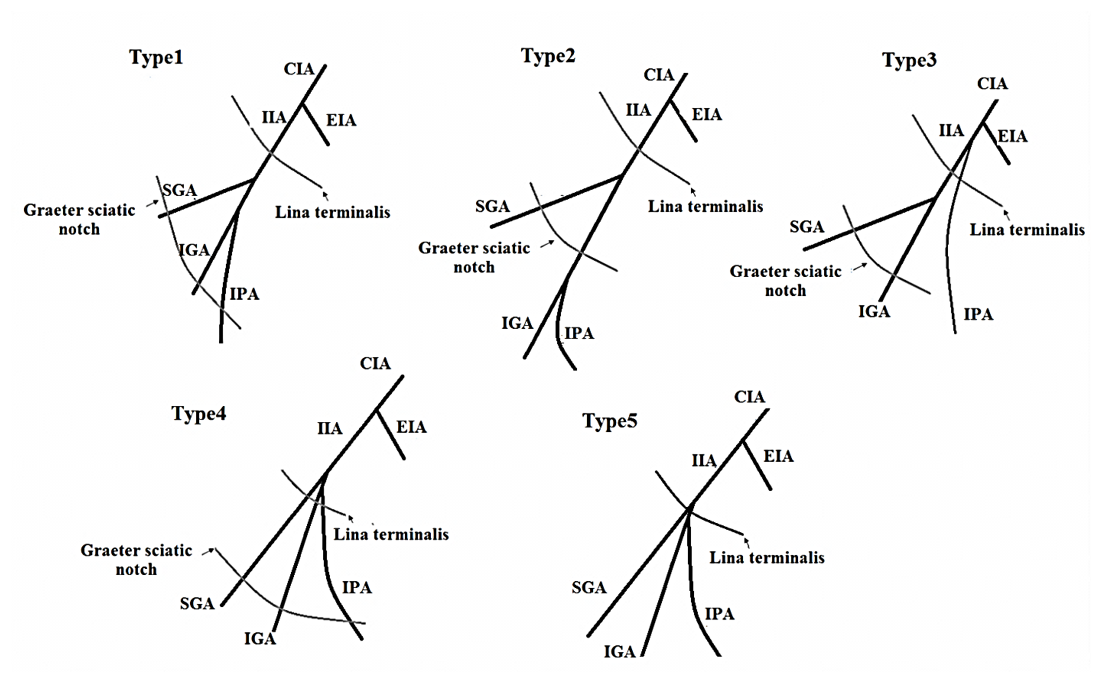
Types of variation the internal pudendal artery (IPA).
Types of variation the internal pudendal artery (IPA) classified by Kawanishi
Types | Variation |
| 1 | The IPA originates from the anterior trunk of IIA at the level between the linea terminalis and the greater sciatic foramen |
| 2 | The IPA originates from the anterior trunk of the IIA at the level of the distal to greater sciatic foramen |
| 3 | The IPA gives off directly from the IIA at a level proximal to the linea terminalis |
| 4 | The IPA, superior and inferior gluteal artery originates from anterior trunk within 1 cm of each other |
| 5 | The penile artery is other than the IPA, such as the obturator artery |
Inferior gluteal artery (IGA)
The IGA is the larger terminal branch of the anterior division of IIA and supplies the buttock and thigh. It descends anteriorly to the piriformis muscle and posterior to the IPA 1. It passes between the piriformis and ischiococcygeus muscle. Then IGA runs through the greater sciatic notch to reach the gluteal region. Gabrielli (1997) showed that the IGA branches penetrated the sciatic nerve in 22.5% of cases30. The IGA and IPA arteries often originate as a common stem from the IIA, sometimes with the SGA (
Origin of inferior gluteal artery (IGA)
Author | Year | Common trunk | Anterior division N (%) | Posterior division N (%) | Absence N (%) |
| Lipshutz | (1916) | Common trunk with IPA (40%) | - | - | - |
| Adachi | (1928) | Common trunk with IPA (51.2%) | - | - | - |
| Braithwaite | (1952) | Common trunk with IPA (60.9%) | - | - | - |
| Roberts & Krishingner | (1968) | Common trunk with IPA (56.4) | - | - | - |
| Bergman | (1988) | Common trunk with the SGA | - | - | - |
| Surekha | (2012) | Case report | absence of IGA | ||
| Reddy | (2007) | Case report | absence of IGA | ||
| Kawanishi | (2008) | Common trunk with the SGA and IPA | - | - | - |
| Nayak | (2012) | - | Case report | - | absence of IGA |
| Havaldar | (2014) | - | Directly in 11 (22%) with IPA in 24 (48%) with ILA in 1 (2%) with OA in 2 (4%) with LSA in 2 (4%) double IGA in 2 (4%) | Directly in 2(4%) with OA in 1 (2%) | 5 (10%) |
| Sakthivelavan | (2014) | Common trunk with IPA (63.2%) | - | - | - |
| Narayana | (2015) | - | 40 (97%) | 1 (3%) | |
| Talalwah | (2015) | Common trunk with IPA and OA (1.5%) | 37.5% (independently with the IPA) 45.7% (dependently with the IPA) | 7.7% | 4.6% |
| Chase & Kirchhoff | (2015) | Common trunk with IPA (48.3) | - | - | - |
| Mohamadi | (2016) | Case report | + | ||
Obturator artery (OA)
One anther of the branches of the anterior trunk of the IIA is obturator artery (OA). This artery passes through the external walls of the pelvis and the artery reaches obturator foramen and enters the obturator canal. In this way it is divided into the vesical, pubic and acetabular branches also, OA supplies the medial compartment of the thigh 1,3. In order to prevent vascular injury and hemorrhage during surgery of the Bogros space and mesh stapling in inguinal or obturator hernia surgery, it is necessary to identify the variations of OA and their distance and orientation to the femoral ring 39. Several papers presented the aberrant of OA in a cross from the pelvic (
Origin of obturator artery (OA)
Origin of obturator artery | Frequencies showed by author in % | |||||||||||
Parsons (1897) | Pick (1942) | Braithwaite (1952) | Pushpa (2006) | Kumar (2007) | Pia (2009) | Havaldar (2014) | Rajive (2015) | Narayana (2015) | Mamatha (2015) | Sonje (2016) | Goke (2016) | |
| Ref. | ||||||||||||
| Common trunk of the IIA | - | - | - | - | - | - | - | 4 | - | - | - | - |
| ILA | 4.9 | 1.8 | 3.5 | - | - | 1.04 | 10 | - | 2 | - | - | - |
| Posterior division of IIA | - | - | - | - | 3.16 | 7.2 | 18 | 10 | - | 14 | 10 | - |
| Anterior division of IIA (normal) | 68.5 | 76 | 75.9 | 100 | - | 61.4 | 40 | 54 | 64 | 66 | 67.9 | - |
| SGA | - | - | - | - | - | 9.3 | - | 2 | - | - | - | - |
| IPA | - | - | - | - | - | - | 8 | 2 | - | - | 10.8 | - |
| Inferior epigastric | 25 | 21.3 | 19.5 | - | - | 14.58 | 6 | 22 | 26 | - | 2 | - |
| Direct form EIA | 1.6 | 0.9 | 1.1 | - | - | 5.2 | 2 | 4 | 8 | 5.1 | 100 (Case report) | |
| By double origin | - | - | - | - | - | - | - | - | - | - | - | |
| IGA | - | - | - | - | - | - | 6 | 2 | - | 4.3 | - | |
| IVA | - | - | - | - | - | - | 4 | - | - | 8 | - | - |
| MRA | - | - | - | - | - | - | 2 | - | - | - | - | - |
| UA | - | - | - | - | - | - | 2 | - | - | - | - | - |
| Abnormal (OA) | - | - | - | - | - | - | - | - | - | 12 | - | - |
| Absent | - | - | - | - | - | - | 2 | - | - | - | - | - |
Visceral branches
Uterine artery (UtA)
Most often, the uterine artery (UtA) is a second or third branch of the anterior division of the IIA. The UtA gives off anterior and posterior arcuate branches which have to anastomose with the ovarian artery. Current literature reportes variant origins for the UtA. These variations were associated with abnormal pregnancy outcome, injury of the artery with ligation or sectioning of the ureter.
Gomez-Jorge (2003) classified the origin of UtA in 4 types 48,49:
Origin of uterine artery (UtA)
Types of variation | Frequencies showed by author in % | |||||
Pelage (1999) | Holub (2005) | Obimbo (2010) | lbulescu (2014) | Havalda (2014) | Mohamadi (2016) | |
| Ref. | ||||||
| 1 | 45% | 30.8 | 18.9% | 24% | 88% | - |
| 2 | 6% | 23.4 | 70.8% | 10% | 12% | - |
| 3 | 43% | 45.6 | 10.4% | 29% | - | - |
| 4 | 6% | - | 0% | 37% | - | +(case report) |
Previous studies documented the umbilical artery (UBA) to be a continuation of the IIA. In embryos, the dorsal aorta of the lower limb divided to ventral (UBA) and dorsal (CIA) branches during the development. Then, the UBA unites with the IIA through vessels anastomosis. The UBA arise from any branch of IIA, such as the main trunk of IIA, SGA, IGA and IPA.
Middle rectal arteries (MRA)
This artery is the visceral brunch of the anterior division of the IIA which is frequently absent. The MRA also stem from the IGA and or IVA with the common trunk at the anterior division of IIA. The role of the MRA is to provide an arterial supply to the muscle of the mid and lower rectum and the extensive rectal anastomosis. Hence, the function of the MRA may be significant for the provision of collateral blood flow during intestinal embolization. Didio (1986) reported that the MRA originated from the IPA in 40% of the studied cases, IGA in26.7% and IIA in16.8. In addition, the MRA was found in 56.7% of the specimens, bilaterally 36.7% and unilaterally 20% 54. Havaldar (2014) described the origins of MRA in 50 specimens as follows: from anterior division (4%), IPA (64%), IVA (6%), OA (2%), IGA (8%). MRA was absent in 16% of cases. One of the complications of surgery procedures such as Hartmann’s procedure, is acupuncture to the proximal rectum region. During the Hartmann, s procedure superior rectal artery is often damaged which due to atrophy of remaining rectal part 8. Naidoo (2018) reported that the MRA was not at principal for the arterial supply of the proximal rectum. However, this can assist to preserve the rectal arterial supply in the procedure of surgery55.
Superior vesical artery (SVA)
According to the anatomical textbook (Gray’s Anatomy), this artery often arises from the anterior division of IIA and UBA, as well as from the OA (4.4%), UtA (9%) and vesicodeferential (9%) (
The SVA supplies the fundus of the bladder, ureter, vas deferens and testis, where it forms an anastomosis with the testicular artery. The beginning of the SVA is at the patent portion of the fetal UBA. The study of Levi and Dubreuil-Chambardel (1925) describe that the number of SVA varies from one to five (
Inferior vesical artery (IVA)
The IVA commonly arises from the anterior division of IIA together with the MRA. IVA may stem from IPA, IGA or together with IPA and SGA (
The vaginal artery (VA)
The VA commonly arises against two or three branches that correspond to the IVA in males. They descend to the vagina and supply the vestibular bulb, vesical fundus, and adjacent part of the rectum.
Origin of Superior vesical artery (SVA)
Origin of SVA | Author | |
Parson and keith (1897) | Bergman (1988) | |
| Ref. | ||
| Hypo gastric trunk | 75.9 | - |
| Anterior division | 15.5 | - |
| IIA | 7 | - |
| MRA | 2 | - |
| Double | 2 | - |
| UtA | - | 4.4 |
| OA | - | 9 |
| vesicodeferential | - | 9 |
Posterior trunk of the IIA
Superior gluteal artery (SGA)
The largest branch of the posterior division of the IIA is the SGA. This artery passes in the vicinity to the lumbosacral plexus and reaches the large sciatic foramen, which then passes through the foramen and out of the pelvis. In 2015, Cook defined the type of arteries according to the pathway of the SGA and its relationship with the lumbosacral trunk (LST) that include 58:
Number of Superior vesical artery
No. of SVA | Author | |
Levi (1902) | Dubreuil-Chambardel (1925) | |
| One | 10% | 9% |
| Two | 70% | 74% |
| Three | 12% | 9% |
| Four | 8% | 6% |
| Five | 8% | 2% |
Origin of inferior vesical artery (IVA) and vaginal artery (VA)
Author | Ref | Origin of IVA or VA | Describe | |
| Bichat (1812) | IIA and UBA | No distinction between men and women | ||
| Kamina (1974, 2014) | IIA and UA | IVA is a male specific artery | ||
| Drake (2015) | UA | IVA No distinction in womenVA is the equivalent of IVA in men | ||
| Moor (2011)Bouchet (1983) | IVA is only in menIVA is replaced in women with VA | |||
| Schunke (2007) | IVA is in women | |||
| Rouviere (2002) | IVA is in two sexes | |||
| Netter (1997) | IIA | IVA is in two sexesVaginal and uterine branch arise from IVA | ||
| Bergman (1988) | Anterior division (22.4%)Hypo gastric trunk (68.9%) | It has anastomosis with UtA in 60-70% of specimen. vaginal artery arises from the UtA, IIA, MRA or SVA | ||
| Havaldar (2014) | Origin of IVA | Anterior division (42%)Double (12%) | It correlate with the research of Bergman | |
| VA | Anterior division (88%)IPA (12%) | |||
Number of lateral sacral arteries (LSA)
Author | Year | Number of LSA% | ||||
One | Two | Three | Four | |||
| Tonkoff | 1898 | 0 | 98 | 2 | 0 | |
| Naguib | 2008 | 30 | 48 | 9 | 0 | |
| Bergman | 50.9 | 47.2 | 0 | 0 | ||
| Sadler | 1990 | 55 | 45 | 0 | 0 | |
| Sharpey | 1867 | 26 | 61.5 | 11 | 1.5 | |
| Poynter | 1922 | 50 | 42.5 | 7.5 | 0 | |
| Talalwa | 2014 | 77.2 | 19.8 | 2.3 | 0.3 | |
| Sakthivelavan | 2014 | 32.7 | 0 | 0 | 0 | |
| Havaldar | 2014 | 24 | 76 | 0 | 0 | |
Origin of lateral sacral arteries (LSA)
Origin of LSA | Frequencies showed by author in % | ||||
Talalwah | Sakthivelavan | Bergman | Bleich | Havaldar | |
| Posterior division | 79.1 | 97.3 | 92.6 | 5.7% | 90 |
| Anterior division | 1.0 | 32.7 | 7.4 | - | 4 |
| SGA | 16.8 | - | - | - | - |
| IGA | 5.4 | - | - | - | - |
| IPA | 0.3 | - | - | - | - |
| Persistent sciatic artery | 8.8 | - | - | - | - |
| Absence | 0.3 | - | - | - | - |
| IIA | 3.7 | - | - | 61.4 | 6 |
Origin of Iliolumbar artery (ILA)
Author | ILA from the CIA | ILA from the bifurcation of the CIA | ILA from the main stem of the IIA | ILA from the point of IIA bifurcation | ILA from the posterior division of the IIA | Ref |
| Chait (1968) | X | |||||
| Hare and Holland (1983) | X | |||||
| Ebraheim (1997) | X 3.7% | X 96.3% | ||||
| Chen (1999) | X | X | ||||
| Mehta (2001) | X | |||||
| Yano (2001) | X | |||||
| Harrington (2001) | X | |||||
| Winters (2002) | X | |||||
| Yiming (2002) | X | |||||
| Elliot and Smit (2006) | X | |||||
| Yoon (2004) | X | X | ||||
| Heye (2006) | X (61%) | X | ||||
| Kiray (2006) | X | |||||
| Bleich (2007) | X (28.3%) | X | ||||
| Naguib (2008) | X (50%) | X (6%) | X (44%) | |||
| Rusu (2010) | 8.75% | 2.5% | 52.5% | 3.75% | 32.5% | |
| Sakthivelavan (2014) | ILA (16.4%)No branch before division (43.1%) | |||||
| Mamatha (2015) | 6% | |||||
| Narayana (2015) | 2% (from OA) |
Lateral sacral arteries (LSA)
Another branch of the posterior trunk of the IIA is the LSA, which is immediately divided into superior and inferior branches after formation. The superior branch passes dorsally to supply the structure of the sacral canal. The inferior branch passes to the ventral surface of piriformis then through the anterior sacral foramen to supply the sacral canal, skin, and muscle over the posterior surface of the sacral region. Several studies show the variation in the origin of the LSA; however, it remains unclear. The current study includes the variability of LSA and its occurrence to provide main anatomical data for radiologists, clinicians and surgeons. In another study, the LSA was found one to four arteries in specimens (1,4) (
Iliolumbar artery (ILA)
The ILA arises from the posterior division of IIA. It runs deep to the EIA and crosses between the obturator nerve and the lumbosacral plexus. ILA then runs between the iliacus and the iliac fascia muscles and supplies muscles and bone. Harrington has reported several branches of ILA that includes: The lumbar, spinal and iliac branches 1,3 (
Conclusion
Identifying and reviewing the locations, orientations, and anatomical details of the IIA is essential for successful performance of endoscopic extraperitoneal inguinal hernioplasty (TEP), ligation of IIA during acute hemorrhage, ureteral injury and vein laceration. Pelvic surgeries may lead to hemorrhage if branching patterns of the IIA are incorrectly interpreted. Moreover, in women, acquiring information on the pelvic vascularization and anatomic variations is necessary for protecting perineal functionality in the case of blood vessels injury. It is important for surgeons and radiologists to have anatomical information about common variations of the uterine artery because these variations may depend on the procedures used to improve hemostasis. Failure to understand the IIA variations can lead to bleeding and thus endanger the patient’s life, exclusively in the severe complications such as ligation of the external iliac artery, removing the prostate, hernia repair or uterine fibroid.
Competing Interests
The authors declare that there is no conflict of interest regarding the contents of this article.
Authors' Contributions
In this study, all authors participated in the various stages of the manuscript editing and approved the final version of manuscript.

