Preliminary evaluation of treatment efficacy of umbilical cord blood-derived mesenchymal stem cell-differentiated cardiac progenitor cells in a myocardial injury mouse model
Abstract
Recently, stem cell therapy has been investigated as a strategy to prevent or reverse damage to heart tissue. Although the results of cell transplantation in animal models and patients with myocardial ischemia are promising, the selection of the appropriate cell type remains an issue that requires consideration. In this study, we aimed to evaluate the effect of cardiac progenitor cell transplantation in a mouse model of myocardial ischemia. The cardiac progenitor cells used for transplantation were differentiated from umbilical cord blood mesenchymal stem cells. Animal models injected with phosphate-buffered saline (PBS) and healthy mice were used as controls. Cell grafting was assessed by changes in blood pressure and histological evaluation. After 14 days of transplantation, the results demonstrated that the blood pressure of transplanted mice was stable, similar to healthy mice, whereas it fluctuated in PBS-injected mice. Histological analysis showed that heart tissue had regenerated in transplanted mice, but remained damaged in PBS-injected mice. Furthermore, trichrome staining revealed that the transplanted mice did not generate a significant amount of scar tissue compared with PBS-injected control mice. In addition, the cardiac progenitor cells managed to survive and integrate with local cells in cell-injected heart tissue 14 days after transplantation. Most importantly, the transplanted cells did not exhibit tumorigenesis. In conclusion, cardiac progenitor cell transplantation produced a positive effect in a mouse model of myocardial ischemia.
Introduction
Over the past half-century, scientific advances in modifying risk factors, drug treatment, and surgical interventions have significantly improved the quality of life and lifespan of patients with damage to the cardiac muscle following myocardial ischemia. However, current treatment strategies fail to reverse scar formation or repair the injured ventricle wall after myocardial infarction (MI), which eventually leads to ventricular dysfunction and arrhythmia. Currently, stem cell therapy is considered as a strategy with great potential to prevent or reverse cardiac muscle damage and promote tissue regeneration.
The preclinical model of ischemic heart disease has raised awareness about the safety and efficiency of stem cell therapy Quyyumi et al., 2011. Despite the promising results of cell transplantation in animal models and patients with ischemic cardiomyopathy, it remains to be determined which cell type is the most appropriate for this therapy. Current cell therapies for heart disease have shown remarkable developments compared with the first cell transplantation performed for heart damage more than 20 years ago Marelli et al., 1992. Initially, scientists focused on using stem cells and skeletal muscle satellite cells because they were easily identified, and exhibited strong proliferation rates and high ischemia-withstanding ability Marelli et al., 1992Murry et al., 2002Murry et al., 1996. However, these cell sources were later shown to be incapable of differentiating into myocardial cells, as well as exhibiting low incorporation with local myocardiocytesin vivo post-transplantation (Murry et al., 2005; Reinecke et al., 2002). Subsequent studies searching for appropriate cell sources capable of inducing positive therapeutic effects examined numerous cell sources, including bone marrow-derived mesenchymal stem cells (MSCs) Mazo et al., 2012. Embryonic stem cells (ESCs) can be differentiated into mature cells and have been shown to regenerate damaged heart tissue in animal models. However, because of bioethical issues, legality (Blum and Benvenisty, 2008), and immunity (Zhu et al., 2009), their use in human trials have been substantially limited to date. In their place, numerous groups have investigated the use of induced pluripotent stem cells (iPSCs) Wernig et al., 2007 or induced cardiomyocytes(Ieda et al.). Recently, the repeatability, reliability, and safety of cell reprogramming and genetic techniques have been examined in clinical trials Kanashiro-Takeuchi et al., 2011Mushtaq et al.,2011. Currently, heart muscle cells derived from adult stem cells, including bone marrow-derived mononuclear cells Hare, 2011Perin et al., 2012Traverse et al., 2009Williams et al., 2011, MSCs Williams and Hare, 2011, adipose tissue stem cells Mazo et al., 2012, and stem cells derived from cardiac tissue Bolli et al., 2011 Makkar et al. have been prioritized for clinical testing and evaluation.
The results of recent clinical trials support the notion that stem cell therapy is safe and is capable of repairing heart structures, as well as restoring heart function Karantalis et al., 2012Williams et al., 2011.
The above results help to confirm the safety and efficiency of MSC transplantation for cardiovascular disease. However, the majority of clinical studies have used autologous bone marrow-derived MSCs. Because the properties of bone marrow-derived MSCs tend to decline with age, studies continuing to examine if alternative cell sources are necessary. Of these cells, human umbilical cord blood-derived stem cells have proven to be a valuable candidate Goldstein et al., 2007. In animal models, cord blood stem cells reduced infarct size, increased heart wall thickness, reduced inflammation at the lesion site, and improved the ejection fraction of the left ventricle Henning et al., 2004Henning et al., 2006Henning et al., 2008. However, before proceeding to clinical studies and applications, further studies are still required.
Until now, the exact mechanism of the effects produced by stem cell transplantation in the treatment of MI remain unknown. However, cell therapy is believed to regenerate myocardial tissue damages through either one or both of the following mechanisms: (i) creating new myocardiocytes and inducing angiogenesis to supply nutrients for myocardial cell mass, and/or (ii) releasing paracrine factors to stimulate cell regeneration, induce the directed differentiation of stem cells, prevent scarring, and stimulate angiogenesis Mazo et al.,2012.
In this study, cardiac progenitor cells, differentiated from human umbilical cord blood-derived MSCs (UCB-MSCs), were transplanted into a mouse model of myocardial ischemia to determine whether they could be induced to differentiate into mature heart muscle cells, which could replace dead tissue and reduce the infarct lesion area.
Materials and Methods
This study was approved by the Institutional Ethical Committee of Laboratory of Stem Cell Research and Application, University of Science, Vietnam National University, Vietnam.
Cell source for transplantation
Third- to fifth-generation human UCB-MSCs were induced to differentiate into CPCs using 5-Azacytidine (5-Aza)-containing medium, according to the process of Pham et al.Pham et al., 2014. Briefly, the cells were induced in Dulbecco’s-modified Eagle’s medium supplemented with 10% fetal bovine serum (FBS); 1% penicillin/streptomycin; 10 μM 5-Aza; 50 ng/mL activin A; 0.1 mM ascorbic acid. After 24 h induction, cells were washed twice with phosphatebuffered saline (PBS) and cultured in Dulbecco’smodified Eagle’s medium supplemented with 15% FBS, 1% penicillin/streptomycin, 50 ng/mL activin A; 0.1 mM ascorbic acid, without 5-Aza. The medium was replaced every 3 days until day 30.
Cell transplantation
Male albino mice (25–30g) were anesthetized by thigh muscle injection with 0.022 g/mL ketamine until the ratio of ketamine anesthetic reached 87 mg/kg of body weight Kanno et al., 2002. Mice were intubated and mechanically ventilated, with pump speed 120–130 cycles/min, pump volume per cycle was 1 mL/100 g body weight. After left thoracotomy, ligation of the left anterior descending artery (LAD) was performed at the distal 1/3 of the coronary artery from the aorta to the heart apex Degabriele et al., 2004. After 3 min, CPCs were infused into the area of infarction, at a density 106 cells per 30 μL for each mouse. Then, the chest was closed, and the muscle layer was sewn tight and applied with antiseptic. The mouse was placed carefully into a clean case. Normal mice were used as positive controls, while LAD ligation plus PBS injection was used as a negative control.
Transplantation efficacy was assessed according to criteria, including blood pressure fluctuation, and the survival, migration, and influence of CPCs on the grafted heart tissue.
Blood pressure monitoring methods
Mice blood pressure changes were monitored using a 58500 Blood Pressure Recorder (Ugo BasileSrl, Italy), 1–2 weeksbefore and after cell transplantation.
To facilitate with stress, mice were trained to be familiar with tail-cuff blood pressure measurements. The animals were anesthetized by placing them in an anesthesia closed chamber, which provided continuous supply of 5% pure oxygen at a rate 1 L/min of isoflurane, until coma was induced. The mouse tail was inserted into the blood pressure measuring ring with signal sensor. Systolic (SYS) and diastolic (DIA) blood pressure of the animals were measured and recorded following the manufacturer’s instructions (58600 Blood Pressure Recorder). Mean arterial pressure (MAP) was calculated by the following formula:
MAP = [(2 × DIA) + SYS] / 3
Heart tissue harvesting and cryosectioning
The animal heart was washed with PBS and fixed in 4% paraformaldehyde overnight. After sucrose cryoprotection, the tissue was moulded with optimum cutting temperature compound and cryosectioned into 10-μm-thick slices. The obtained slices were then treated with hematoxylin and eosin (H&E) stain, Trichrome stain, and immunohistochemistry.
Hematoxylin & eosin staining
H&E staining was performed to evaluate tissue and cell structures. Slices were immersed in a graded series of alcohol solutions as follows: 100% for 2 min, 90% for 2 min, 70% for 1 min, 50% for 1 min, and double- distilled water for 2 min. The surrounding tissue area was dried with absorbent paper, followed by an addition of a drop of hematoxylin stain onto the tissue slice. After 2 min, the slice was washed with distilled water. Two drops of eosin stain were added to the tissue slice for 30 s and the specimen was successively embedded again with: 50% alcohol for 1 min, 70% alcohol for 1 min, 90% alcohol for 1 min, and 100% alcohol for 1 min. Immersion oil was dispensed onto the slice followed by lamelle fixation.
Trichrome staining
Masson’s Trichromedye is commonly used to distinguish collagen in pathological cases. Three dyes are used to identify collagen, fibrin, and erythrocytes. Tissue was initially stained with Biebrich Scarlet acidic dye (light red), which has a binding preference to acidic components in the tissue. When the tissue was treated with phosphomolybdic acid, the red color was displaced from the collagen component. In addition, phosphomolybdic acid also generated links between collagen and light green molecules that tainted the collagen green.
Tissue specimens were immersed in a graded series of alcohol solutions as follows: 100% for 1 min, 90% for 1 min, and 80% for 1 min. Samples were washed gently twice with distilled water, followed by immersion in Bouin-containing solution overnight at 56 °C. Samples were rinsed with water to remove traces of piric acid (yellow) and soaked in Weigert’s iron hematoxylin solution for 10 min. Samples were gently washed continuously with distilled water for 30 s. Next, they were embedded in Biebrich scarlet solution for 1 min. The slices were washed quickly again with distilled water and immersed in 5% phosphomolybdic acid solution for 30 min. Then, samples were transferred and bathed in the light green solution for 10 min. The samples were rinsed quickly with distilled water and immersed in 0.5% acetic acid solution for 2 min. The samples were immersed in a series of graded alcohol solutions as follows: 95% for 1 min × 2, followed by 100% for 1 min × 2. Immersion oil was dropped onto the slices, followed by lamelle fixation. Trichromestained sections were identified and collagen structures were evaluated by ImageJ software.
Immunocytochemistry staining
Cells grown on coverslips (Santa Cruz Biotechnology, Dallas, TX, USA) were prepared for immunocytochemistry as follows: cells were fixed in % paraformaldehyde solution for 15 min; permeabilized with 0.1% Triton X-100; washed three times in PBS; blocked with BSA; and incubated with a primary human antibody alpha-actinin (Abcam 1:400) or human Troponin T (Abcam 1:400) overnight at 4 °C. After washing, the samples were treated with goat anti-rabbit IgG secondary antibodies (Abcam 1:400) and Hoechst 33342 (Sigma 1:400) for 45 min at room temperature. The samples were rinsed three times with PBS, mounted, and observed under a fluorescent microscope (Zeiss Axiovert).
Immunohistochemistry staining
Tissue sections were fixed and stained with Hoechst 33342 (Sigma, 1:400) and antibodies specific for human proteins of interest, Troponin T (Abcam, 1:200) and alpha-actinin (Abcam, 1:200), according to the manufacturer’s protocol. Slides containing tissue sections were washed briefly for 2 × 5 min in PBS. Tissue sections were blocked with 5% BSA at room temperature for 30 min and incubated with primary antibodies at room temperature for 60 min. After washing, slides were then treated with secondary antibodies at room temperature for 60 min in the absence of light to avoid optical bleaching. The samples were washed 4 × 5 min in PBS, coated with polyvinyl alcohol solution, and covered by lamelle. Their images were observed and recorded by fluorescence microscopy.
Reverse transcription-polymerase chain reaction
Before transplantation, CPCs were tested for the expression of human Sox2 and Oct4 genes, which are used to characterize tumor formation, using reverse transcription-polymerase chain reaction (RT-PCR). Human glyceraldehyde 3-phosphate dehydrogenase (hGAPDH) was used as an internal control.
At 14 days post-transplantation, total RNA from hearts of control animals or test animals was collected to examine the tumor formation capacity of infused cells in vivo.
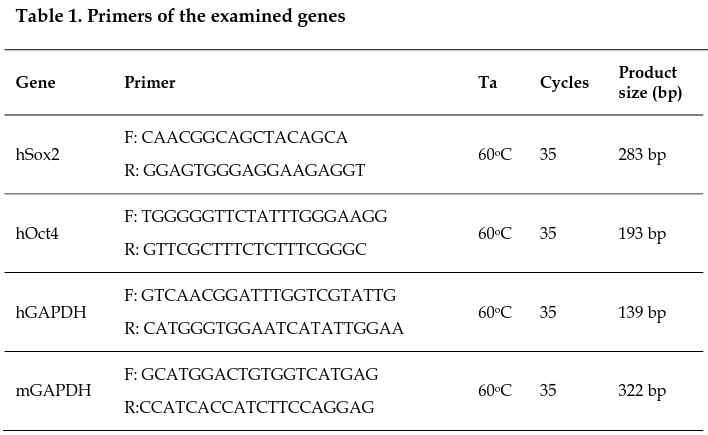
Total RNA was extracted from CPCs prepared for transplantation, human MCF7 breast cancer cells, and heart tissue samples 14 days post-transplantation using Easy Blue Reagent (Intron), following the manufacturer’s instructions. cDNA was synthesized from RNA using the Superscript II Reverse Transcriptase (Invitrogen), according to the manufacturer’s procedure. Next, PCR was performed using Taq DNA Polymerase (Takara, Shiga, Japan). The primers of the examined genes are listed in Table 1 .
Gel electrophoresis was performed on RT-PCR products on 2% agarose gels, at 100 V, for 60 min. A 100-bp DNA ladder was used for all products. The results were analyzed by electrophoresis using the Gel Doc IT system (UVP).
Statistical Analysis
Data were statistically interpreted using GraphPad Prism software version 6.
Results
UCB-MSC differentiation into CPCs
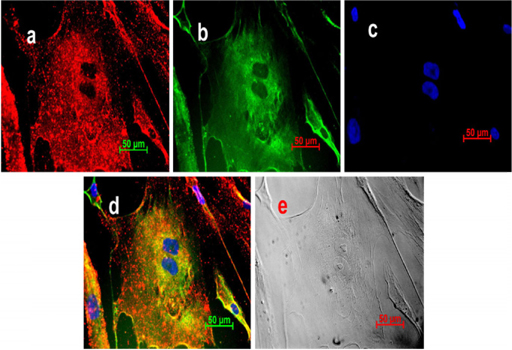
UCB-MSCs were induced in medium containing 5-Aza. The induced cells were checked by ICC staining with antibodies specific for human cardiac muscle cells, including human alpha-actinin and human troponin T. The results indicated that a phenotypic change occurred in the induced cells. Originally, the population exhibited the morphology of MSCs, which are mononuclear and spindle shaped; then, they transformed into multinuclear cells, which are similar to those of cardiac muscle cell lineages. Additionally, they also expressed both specific markers of cardiac muscle cells, human alpha-actinin and human troponin T ( Figure 1 ).
Blood pressure changes
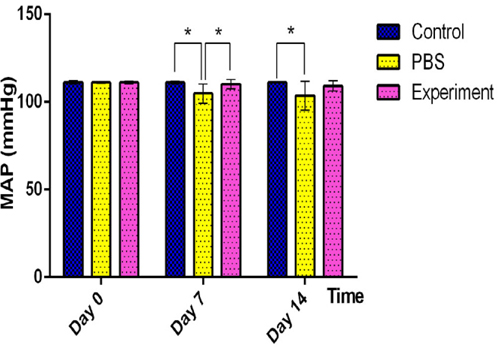
To assess the effect of cell transplantation in the mouse model of myocardial ischemia, changes in blood pressure before and after transplantation were recorded through a dedicated system ( Figure 2 ). The collected data were processed using statistical software.
In normal mice, the MAP was approximately 111 mmHg Tiemann et al., 2003. Healthy mice (n = 10) were used as a positive control with stable blood pressure (DO = 111.1 mmHg; 07 = 111.0 mmHg; 014 = 111.1 mmHg). However, MAP in the LAD ligation and PBS-injected mice (PBS group) (n = 10) tended to decrease over the survey period (DO = 111.0 mmHg; 07 = 104.8 mmHg; 014 = 103.5 mmHg). This result suggested that heart activity was decreased in the PBS group. Meanwhile, the MAP in the CPC-transplanted group (n = 10) was decreased compared with the control group (DO = 111.0 mmHg; 07 = 110.1 mmHg; 014 = 109.1 mmHg); however, the decrease was evidently less than that of the PBS group.
CPC transplantation reduced heart tissue damages
To assess the effects of grafted CPCs in the mouse model, animals were divided into three groups. The control group consisted of normal healthy mice; the PBS group consisted of mice receiving LAD ligation and PBS injection; and the CPC group comprised mice receiving LAD ligation and CPC transplantation.
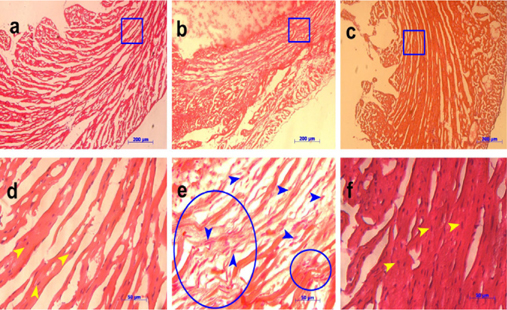
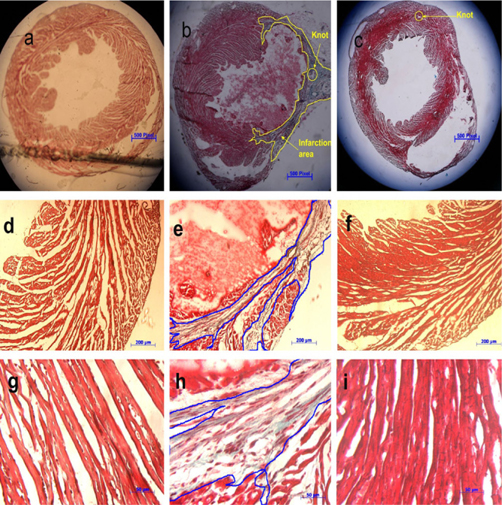
The heart tissue was fixed and cryo-sectioned onto slides (lO-f-lm-thick slices). Slides were then stained with H&E for tissue damage assessment. Results showed that heart tissue in the control group remained intact and exhibited no sign of inflammation. The left ventricle wall of the control group was normal, while it was thinner in the PBS group. Heart tissue of the PBS group exhibited more loss of crossstriations, with significantly shrunken bands and nuclear karyolysis in heart cells (green arrow, Figure 3 ). Some neutrophils infiltrated the heart muscle and fibers appeared rippled in the PBS group (area circled in blue, Figure 3 ). However, no fracture or damage of the heart tissue was observed on the CPC group. In addition, the nucleus was located in the center of the cell (yellow arrows, Figure 3 ), pale pink plates were also observed and the left ventricle wall was not as thin as the PBS group ( Figure 3 ).
To assess scar formation in the damaged heart tissue and effects of CPCs on the process, mice hearts were collected for Trichrome staining. Results showed that the myocardium of the control group did not experience fibrosis ( Figure 4a , Figure 4d , Figure 4g ), while the PBS group formed broad blue regions of fibrosis when stained with Trichrome ( Figure 4b , Figure 4e , Figure 4h ). No sign of fibrosis was observed in the myocardium of the CPC group compared with the PBS group ( Figure 4c , Figure 4f , Figure 4i ).
Post-transplantation evaluation of infused CPCs in heart tissue
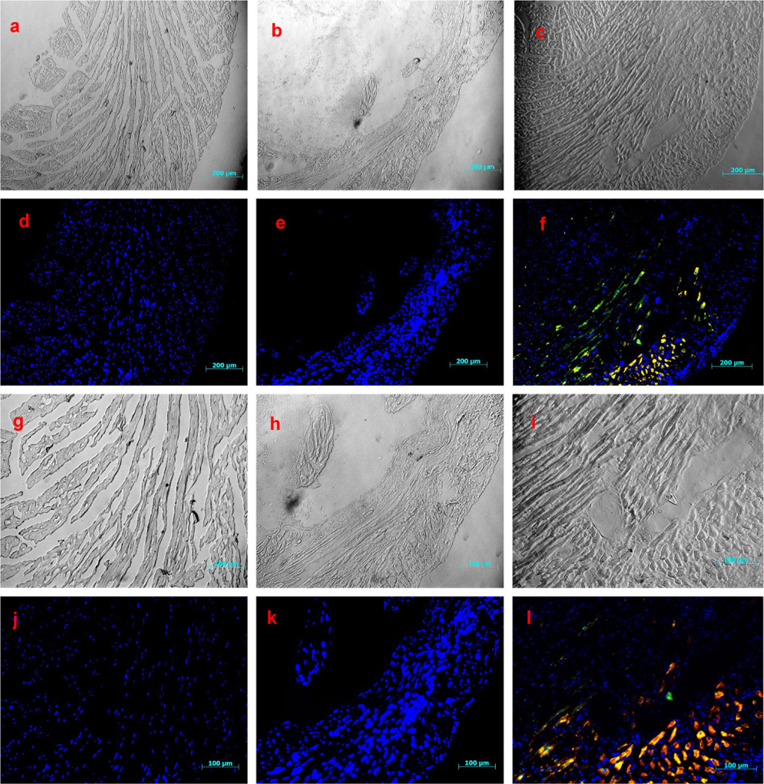
To investigate the fate of the infused CPCs in the damaged heart tissue of experiment animals, mouse heart tissue samples were collected, frozen, and sectioned. Slices were then stained with antibodies, which were specific for human heart cells, including human alpha-actinin and human troponin T.
The results of tissue staining indicated that heart tissues of the control and PBS groups were negative for staining with these specific antibodies ( Fig. 5a, d, g, j, b, e, h, k ). Meanwhile, some areas of CPC-transplanted heart tissue exhibited positive staining ( Fig. 5c, f, i, l ), indicating that grafted CPCs survived and expressed both human Troponin T (green) and human alpha-actinin (red). These cells not only did not form scar tissue but they also prevented or reduced tissue damage caused by ischemia. Therefore, the left ventricular walls of CPC-transplanted mice were relatively thick compared with normal mice. In contrast, mice in the PBS group exhibited thin ventricular walls and scar formation.
CPCs did not induce tumorigenesis post-transplantation
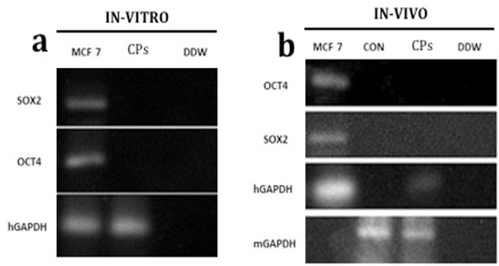
To test for the tumor-forming ability of the transplanted cells prior to transplantation, CPCs were evaluated for human cancer gene expression, including human Sox2 (hSox2) and human Oct4 (hOct4). The human breast cancer MCF7 cell line was used as a positive control ( Figure 6 ) and distilled water was used as a negative control. The results showed that the CPCs did not express hOct4 and hSox2 compared with MCF7 cells ( Figure 7a ).
To assess whether CPC survival and tumorigenesis in mouse cardiac tissue post-transplantation, CPC-transplanted tissue was collected. Total RNA was extracted from heart tissue for further analysis of hSox2 and hOct4 expression. RNA from normal mouse heart was used for comparison. MCF7 cells and distilled water controls were treated with the same procedure. The results showed that transplanted CPCs existed in grafted tissues and the tumor genes hSox2 and hOct4 were not expressed ( Figure 7b ).
Discussion
In this study, we evaluated the potential of CPCs to repair damaged myocardial tissue. CPC transplantation minimized blood pressure fluctuation in transplanted mice similar to the control group; and was more stable compared with the PBS group. The engrafted CPCs survived and integrated with the host cardiac tissue, improved tissue structure, and prevented the progression of fibrosis in the damaged heart muscle 14 days after transplantation. CPC transplantation did not induce teratoma formation and the expression of cancer genes, including hOct4 and hSox2, in CPC-transplanted mice. In other studies, the transplantation of undifferentiated ESCs into rat models of MI tended to generate teratomas Nussbaum et al., 2007. In our study, the transplantation of CPCs derived from UCB-MSCs did not form teratomas in transplanted tissue ( Figure 6 , Figure 7 ). This difference may be a result of the induction of different changes in the gene expression of CPCs and ESCs, whereby CPCs did not undergo oncogenic differentiation. This result was similar to the previous results of Huber et al., where human ESC-derived cardiomyocytes (hESC-CMs) were infused into a mouse model of MI Huber et al., 2007. The transplantation of these cells after MI did not lead to the formation of teratomas. However, the engrafted cardiomyocytes also failed to survive, proliferate, or integrate with host cardiac tissue Huber et al., 2007Klug et al., 1996. In this study, the transplanted CPCs survived and integrated with the host cells ( Figure 5 ), which could be related to differences in cell sources, monitoring time, and cell differentiation procedures. However, hESC-CM transplantation improved damaged heart function in a similar manner as CPC transplantation. Because cardiomyocyte loss plays a significant role in the development of heart failure, cardiomyogenesis is thus an important process in the regeneration of heart tissue Laflamme and Murry, 2005, 2011. Interestingly, the results of CPC transplantation after 14 days showed that the left ventricular wall of the CPC-infused group was thicker compared with the PBS group ( Figure 3 ). Trichrome staining results also confirmed the absence of scar formation from the heart tissue of the CPC group ( Figure 4 ), while a large scar was observed in the heart tissue of the PBS group ( Figure 4 ). This result suggested that grafted CPCs improved cardiac function deterioration in damaged heart tissue. Currently, there are several theories on how cell transplantation improves myocardial perfusion and cardiac function. The transplanted cells are capable of differentiating into myocardial cells after infusion and eventually improve cardiac function. In addition, the transplanted cells may possess the ability to release paracrine factors, such as vascular endothelial growth factor, insulin-like growth factor, hepatocyte growth factor, and fibroblast growth factor, which activate endogenous cardiac progenitors to protect and repair lost heart muscle. In this study, it was demonstrated that the delivered CPCs managed to survive in the grafted tissue and did not transform into muscle fibers, but maintained the functions of cardiac cells ( Figure 4 , Figure 5 ). In contract, the heart tissue of the PBS group was severely damaged ( Figure 4 , Figure 5 ). This result was similar to previous studies about the ability of cells to integrate with host cardiac tissue both in vitro and in vivo Kehat et al., 2004Xue et al., 2005. This effect was identical to the positive effect of menstrual bloodderived MSC (MMC) transplantation 2 weeks after LAD ligation Hida et al., 2008; or similar to the results of porcine iPSC transplantation in a MI model Li et al., 2013. The similarity between the grafted MMCs and iPSCs was that both exhibited the ability to differentiate into heart cells after transplantation and displayed positive effects in clinical trials. Conversely, cardiac MSCs formed a scar after transplantation Carlson et al., 2011Silva et al.,2014, hESCs formed teratomas after transplantation Hentze et al., 2009, and BM-MSCs had no influential effects on the grafted heart tissue after infarction Grauss et al., 2007Hou et al., 2007. Meanwhile, these cell types lack the ability to differentiate into heart muscle cells after transplantation, whereas grafted CPCs were positive for specific antibodies of myocardial cells before and after transplantation, which might explain why CPC transplantation was shown to have a favorable effect in animal models compared with other cell types. Although the initial results were encouraging, as well as showing significant potential for CPC application in animal models of damaged heart tissue caused by ischemia, further studies should be conducted using alternative methods, such as electrocardiography, or by increasing the experimental evaluation timeline to produce stronger results.
Conclusion
The CP transplantation therapy in the mouse model of damaged myocardial tissue caused by ischemia initially showed positive effects for the prevention of myocardial injury after LAD ligation.
Ethical standard statements
Umbilical cord blood samples collected from informed consent of the mother used to isolate MSCs. The collection was performed in accordance with the ethical standards of the local ethics committee Van Hanh General Hospital, Ho Chi Minh city, Vietnam. All procedures and manipulationsof animals have been approved by the Institutional Ethical Committee of Laboratory of Stem Cell Research and Application, University of Science, Vietnam National University, Vietnam.
References
-
B.
Blum,
N.
Benvenisty.
The tumorigenicity of human embryonic stem cells. Advances in cancer research.
2008;
100
:
133-158
.
-
R.
Bolli,
A.R.
Chugh,
D.
D'Amario,
J.H.
Loughran,
M.F.
Stoddard,
S.
Ikram,
G.M.
Beache,
S.G.
Wagner,
A.
Leri,
T.
Hosoda.
Cardiac stem cells in patients with ischaemic cardiomyopathy (SCIPIO): initial results of a randomised phase 1 trial. Lancet (London , England).
2011;
378
:
1847-1857
.
-
S.
Carlson,
J.
Trial,
C.
Soeller,
M.L.
Entman.
Cardiac mesenchymal stem cells contribute to scar formation after myocardial infarction. Cardiovascular research.
2011;
91
:
99-107
.
-
N.M.
Degabriele,
U.
Griesenbach,
K.
Sato,
M.J.
Post,
J.
Zhu,
J.
Williams,
P.K.
Jeffery,
D.M.
Geddes,
E.W.
Alton.
Critical appraisal of the mouse model of myocardial infarction. Experimental physiology.
2004;
89
:
497-505
.
-
G.
Goldstein,
A.
Toren,
A.
Nagler.
Transplantation and other uses of human umbilical cord blood and stem cells. Current pharmaceutical design.
2007;
13
:
1363-1373
.
-
R.W.
Grauss,
E.M.
Winter,
J.
van Tuyn,
D.A.
Pijnappels,
R.V.
Steijn,
B.
Hogers,
R.J.
van der Geest,
A.A.
de Vries,
P.
Steendijk,
A.
van der Laarse.
Mesenchymal stem cells from ischemic heart disease patients improve left ventricular function after acute myocardial infarction. American journal of physiology Heart and circulatory physiology.
2007;
293
:
H2438-2447
.
-
J.M.
Hare.
Bone marrow therapy for myocardial infarction. Jama.
2011;
306
:
2156-2157
.
-
R.J.
Henning,
H.
Abu-Ali,
J.U.
Balis,
M.B.
Morgan,
A.E.
Willing,
P.R.
Sanberg.
Human umbilical cord blood mononuclear cells for the treatment of acute myocardial infarction. Cell transplantation.
2004;
13
:
729-739
.
-
R.J.
Henning,
J.D.
Burgos,
L.
Ondrovic,
P.
Sanberg,
J.
Balis,
M.B.
Morgan.
Human umbilical cord blood progenitor cells are attracted to infarcted myocardium and significantly reduce myocardial infarction size. Cell transplantation.
2006;
15
:
647-658
.
-
R.J.
Henning,
M.
Shariff,
U.
Eadula,
F.
Alvarado,
M.
Vasko,
P.R.
Sanberg,
C.D.
Sanberg,
V.
Delostia.
Human cord blood mononuclear cells decrease cytokines and inflammatory cells in acute myocardial infarction. Stem cells and development.
2008;
17
:
1207-1219
.
-
H.
Hentze,
P.L.
Soong,
S.T.
Wang,
B.W.
Phillips,
T.C.
Putti,
N.R.
Dunn.
Teratoma formation by human embryonic stem cells: evaluation of essential parameters for future safety studies. Stem cell research.
2009;
2
:
198-210
.
-
N.
Hida,
N.
Nishiyama,
S.
Miyoshi,
S.
Kira,
K.
Segawa,
T.
Uyama,
T.
Mori,
K.
Miyado,
Y.
Ikegami,
C.
Cui.
Novel cardiac precursor-like cells from human menstrual blood-derived mesenchymal cells. Stem cells.
2008;
(Dayton
:
Ohio) 26, 1695-1704
.
-
M.
Hou,
K.M.
Yang,
H.
Zhang,
W.Q.
Zhu,
F.J.
Duan,
H.
Wang,
Y.H.
Song,
Y.J.
Wei,
S.S.
Hu.
Transplantation of mesenchymal stem cells from human bone marrow improves damaged heart function in rats. International journal of cardiology.
2007;
115
:
220-228
.
-
I.
Huber,
I.
Itzhaki,
O.
Caspi,
G.
Arbel,
M.
Tzukerman,
A.
Gepstein,
M.
Habib,
L.
Yankelson,
I.
Kehat,
L.
Gepstein.
Identification and selection of cardiomyocytes during human embryonic stem cell differentiation. FASEB journal : official publication of the Federation of American Societies for Experimental Biology.
2007;
21
:
2551-2563
.
-
M.
Ieda,
J.-D.
Fu,
P.
Delgado-Olguin,
V.
Vedantham,
Y.
Hayashi,
B.G.
Bruneau,
D
Srivastava.
Direct Reprogramming of Fibroblasts into Functional Cardiomyocytes by Defined Factors. Cell.
;
142
:
375-386
.
-
R.M.
Kanashiro-Takeuchi,
I.H.
Schulman,
J.M.
Hare.
Pharmacologic And Genetic Strategies To Enhance Cell Therapy For Cardiac Regeneration. Journal of molecular and cellular cardiology.
2011;
51
:
619-625
.
-
S.
Kanno,
D.L.
Lerner,
R.B.
Schuessler,
T.
Betsuyaku,
K.A.
Yamada,
J.E.
Saffitz,
A.
Kovacs.
Echocardiographic evaluation of ventricular remodeling in a mouse model of myocardial infarction. Journal of the american society of echocardiography.
2002;
15
:
601-609
.
-
V.
Karantalis,
W.
Balkan,
I.H.
Schulman,
K.E.
Hatzistergos,
J.M.
Hare.
Cell-based therapy for prevention and reversal of myocardial remodeling. American journal of physiology Heart and circulatory physiology.
2012;
303
:
H256-270
.
-
I.
Kehat,
L.
Khimovich,
O.
Caspi,
A.
Gepstein,
R.
Shofti,
G.
Arbel,
I.
Huber,
J.
Satin,
J.
Itskovitz-Eldor,
L.
Gepstein.
Electromechanical integration of cardiomyocytes derived from human embryonic stem cells. Nature biotechnology.
2004;
22
:
1282-1289
.
-
M.G.
Klug,
M.H.
Soonpaa,
G.Y.
Koh,
L.J.
Field.
Genetically selected cardiomyocytes from differentiating embronic stem cells form stable intracardiac grafts. The Journal of clinical investigation.
1996;
98
:
216-224
.
-
M.A.
Laflamme,
C.E.
Murry.
Regenerating the heart. Nature biotechnology.
2005;
23
:
845-856
.
-
M.A.
Laflamme,
C.E.
Murry.
Heart regeneration. Nature.
2011;
473
:
326-335
.
-
X.
Li,
F.
Zhang,
G.
Song,
W.
Gu,
M.
Chen,
B.
Yang,
D.
Li,
D.
Wang,
K.
Cao.
Intramyocardial Injection of Pig Pluripotent Stem Cells Improves Left Ventricular Function and Perfusion: A Study in a Porcine Model of Acute Myocardial Infarction. PloS one.
2013;
8
:
e66688
.
-
R.R.
Makkar,
R.R.
Smith,
K.
Cheng,
K.
Malliaras,
L.E.J.
Thomson,
D.
Berman,
L.S.C.
Czer,
L.
Marbán,
A.
Mendizabal,
P.V.
Johnston.
Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): a prospective, randomised phase 1 trial. The Lancet.
;
379
:
895-904
.
-
D.
Marelli,
C.
Desrosiers,
M.
el-Alfy,
R.L.
Kao,
R.C.
Chiu.
Cell transplantation for myocardial repair: an experimental approach. Cell transplantation.
1992;
1
:
383-390
.
-
M.
Mazo,
M.
Araña,
B.
Pelacho,
F.
Prosper.
Mesenchymal Stem Cells and Cardiovascular Disease: A Bench to Bedside Roadmap. Stem Cells International.
2012;
2012
:
11
.
-
C.E.
Murry,
L.J.
Field,
P.
Menasche.
Cell-based cardiac repair: reflections at the 10-year point. Circulation.
2005;
112
:
3174-3183
.
-
C.E.
Murry,
M.L.
Whitney,
H.
Reinecke.
Muscle cell grafting for the treatment and prevention of heart failure. Journal of cardiac failure.
2002;
8
:
S532-541
.
-
C.E.
Murry,
R.W.
Wiseman,
S.M.
Schwartz,
S.D.
Hauschka.
Skeletal myoblast transplantation for repair of myocardial necrosis. Journal of Clinical Investigation.
1996;
98
:
2512-2523
.
-
M.
Mushtaq,
B.N.
Oskouei,
J.M.
Hare.
Cell therapy for heart disease: to genetically modify or not, that is the question. Circulation research.
2011;
108
:
398-401
.
-
J.
Nussbaum,
E.
Minami,
M.A.
Laflamme,
J.A.
Virag,
C.B.
Ware,
A.
Masino,
V.
Muskheli,
L.
Pabon,
H.
Reinecke,
C.E.
Murry.
Transplantation of undifferentiated murine embryonic stem cells in the heart: teratoma formation and immune response. FASEB journal : official publication of the Federation of American Societies for Experimental Biology.
2007;
21
:
1345-1357
.
-
E.C.
Perin,
J.T.
Willerson,
C.J.
Pepine,
T.D.
Henry,
S.G.
Ellis,
D.X.
Zhao,
G.V.
Silva,
D.
Lai,
J.D.
Thomas,
M.W.
Kronenberg.
Effect of transendocardial delivery of autologous bone marrow mononuclear cells on functional capacity, left ventricular function, and perfusion in chronic heart failure: the FOCUS-CCTRN trial. Jama.
2012;
307
:
1717-1726
.
-
T.L.
Pham,
T.T.
Nguyen,
A.
Van Bui,
M.T.
Nguyen,
P.
Van Pham.
Fetal heart extract facilitates the differentiation of human umbilical cord blood-derived mesenchymal stem cells into heart muscle precursor cells. Cytotechnology.
2014
.
-
A.A.
Quyyumi,
E.K.
Waller,
J.
Murrow,
F.
Esteves,
J.
Galt,
J.
Oshinski,
S.
Lerakis,
S.
Sher,
D.
Vaughan,
E.
Perin.
CD34(+) cell infusion after ST elevation myocardial infarction is associated with improved perfusion and is dose dependent. American heart journal.
2011;
161
:
98-105
.
-
H.
Reinecke,
V.
Poppa,
C.E.
Murry.
Skeletal muscle stem cells do not transdifferentiate into cardiomyocytes after cardiac grafting. Journal of molecular and cellular cardiology.
2002;
34
:
241-249
.
-
D.N.
Silva,
B.S.
de Freitas Souza,
C.M.
Azevedo,
J.F.
Vasconcelos,
R.H.
Carvalho,
M.B.
Soares,
R.R.
Dos Santos.
Intramyocardial transplantation of cardiac mesenchymal stem cells reduces myocarditis in a model of chronic Chagas disease cardiomyopathy. Stem cell research & therapy.
2014;
5
:
81
.
-
K.
Tiemann,
D.
Weyer,
P.C.
Djoufack,
A.
Ghanem,
T.
Lewalter,
U.
Dreiner,
R.
Meyer,
C.
Grohe,
K.B.
Fink.
Increasing myocardial contraction and blood pressure in C57BL/6 mice during early postnatal development. American journal of physiology Heart and circulatory physiology.
2003;
284
:
H464-474
.
-
J.H.
Traverse,
T.D.
Henry,
D.E.
Vaughan,
S.G.
Ellis,
C.J.
Pepine,
J.T.
Willerson,
D.X.
Zhao,
L.B.
Piller,
M.S.
Penn,
B.J.
Byrne.
Rationale and design for TIME: A phase II, randomized, double-blind, placebo-controlled pilot trial evaluating the safety and effect of timing of administration of bone marrow mononuclear cells after acute myocardial infarction. American heart journal.
2009;
158
:
356-363
.
-
M.
Wernig,
A.
Meissner,
R.
Foreman,
T.
Brambrink,
M.
Ku,
K.
Hochedlinger,
B.E.
Bernstein,
R.
Jaenisch.
In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature.
2007;
448
:
318-324
.
-
A.R.
Williams,
J.M.
Hare.
Mesenchymal stem cells: biology, pathophysiology, translational findings, and therapeutic implications for cardiac disease. Circulation research.
2011;
109
:
923-940
.
-
A.R.
Williams,
B.
Trachtenberg,
D.L.
Velazquez,
I.
McNiece,
P.
Altman,
D.
Rouy,
A.M.
Mendizabal,
P.M.
Pattany,
G.A.
Lopera,
J.
Fishman.
Intramyocardial stem cell injection in patients with ischemic cardiomyopathy: functional recovery and reverse remodeling. Circulation research.
2011;
108
:
792-796
.
-
T.
Xue,
H.C.
Cho,
F.G.
Akar,
S.Y.
Tsang,
S.P.
Jones,
E.
Marban,
G.F.
Tomaselli,
R.A.
Li.
Functional integration of electrically active cardiac derivatives from genetically engineered human embryonic stem cells with quiescent recipient ventricular cardiomyocytes: insights into the development of cell-based pacemakers. Circulation.
2005;
111
:
11-20
.
-
W.-Z.
Zhu,
K.
Hauch,
C.
Xu,
M.A.
Laflamme.
Human Embryonic Stem Cells and Cardiac Repair. Transplantation reviews.
2009;
(Orlando , Fla)
:
23, 53-68
.
Comments
Downloads
Article Details
Volume & Issue : Vol 2 No 12 (2015)
Page No.: 435-445
Published on: 2015-12-25
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 8335 times
- Download PDF downloaded - 1726 times
- View Article downloaded - 4 times
 Biomedpress
Biomedpress